Category: products
Showing 121–160 of 332 results
-
-
-
CVC Single Lumen 14g*8cm
These catheters offer more prominent implantation rates and upgraded over the wire situation. The plastic clasps are anything but difficult to handle and crimp safe. The offered scope of Central Venous Catheter Single Lumen Kit is accessible in French measurements furthermore in altered lengths for unique techniques.
Features- These are designed with extreme precision
- These are easy to operate and safe to use
- The product comes with assured guarantee
- These are available in a competitive price
-
DELIVERY SYSTEM 6F 18080
- Built-in nebulizer kit holder for hands-free use
- Delivers consistent particle size of <5 microns and fast treatment times
- Complete with disposable nebulizer kit with mouthpiece, T and tubing
- Powerful piston pump is efficient, dependable and quiet.
These size is also available-
DELIVERY SYSTEM 7F 180/80 DELIVERY SYSTEM 7F 45/80 DELIVERY SYSTEM 8F 45/80 -
DIAGNOSTIC CATHETER
Cordis Infiniti Diagnostic
Catheter
The Cordis INFINITI 4F, 5F and 6F line of diagnostic catheters is ideal for coronary angioplasty. These catheters incorporate proprietary Vestan Nylon to deliver exceptional responsiveness and flow rates, optimal torque, and shape retention.
Description
Hydrophilic Coated Guidewires
Regular and stiff hydrophilic wires for access and interventional procedures:
• facilitates access to the most tortuous anatomy
• providing the control, visibility and exceptional design symmetry
• enables to torque and maneuver through difficult lesionsProduct Benefits:
Easy navigation
• Increased turn-for-turn responsiveness
• leading to multivessel selection and lesion crossability
Greater visibility
• Excellent radiopacity can help promote faster and more accurate procedures
Lasting lubricity
• Access with excellent guide and device support, with a smooth ride from start to finish -
Dialyzer f*8
Fresenius Dialyzer F 6 F 8 High Flux Low Flux .The proven technology of Fresenius Polysulfone® and its exceptional quality, biocompatibility and performance provide the foundation for the Hemoflow dialyzer line. The Hemoflow line provides high flux, low flux, single use, reuse and low volume dialyzers. Sterilization methods include Ethylene Oxide (EtO), electron beam (E beam) and steam. The diversity of product options in the Hemoflow line make it an ideal choice for specific patient requirements.”
-
DILATOR ACCURA
Accura: Accura Balloon Catheter PTMC BALLOON
- The Accura Double Lumen PTMC balloon dilation catheter has a 11 F, 80 cm shaft and a three stage expandable balloon
- The balloon is well designed for the safe and optimal expansion of the mitral valve
- Catheter tip design ensures rapid and smooth entry into the mitral valve
- Balloon design ensures rapid inflation and deflation
- Optimum compliance of the balloon material ensures even dilation of the mitral valve
- The Accura Balloon is a dual lumen balloon and does not have a vent tube
- No hole on the outside of the balloon ensures no seepage of blood when balloon is fully expanded
These sizes are also available-
LA WIRE ACCURA METAL STRETCHING TUBE PTMC 23-26 PTMC 25-28 RULER STYLET SYRINGE -
DISPOSABLE FACE MASK
Disposable Face Mask.
Features:
- Skin friendly
- Light-weight
- Comfortable to wear
-
DUODERM XTHIN
Designed to reduce the risk of further skin breakdown due to friction.
DuoDERM® Extra Thin Dressing can be used as a primary hydrocolloid dressing for dry to lightly exuding wounds.
It can be used as a secondary dressing to secure an AQUACEL® Dressing or an AQUACEL® Ag Dressing.
The European Pressure Ulcer Advisory Panel (EPUAP) and The National Pressure Ulcer Advisory Panel (NPUAP) guidelines recommend the usage of hydrocolloids for the management of pressure ulcers.
DuoDERM® Extra Thin Dressing can be used to manage stage I and stage II pressure ulcers. -
DXTERITY DIAGNOSTIC CATHETER
DxTerity™
Diagnostic CatheterPRODUCT DETAILS†
DxTerity Catheters Feature:
- Unique, ergonomic, winged hubs for easy manipulation
- Solid shaft construction for efficient torque and excellent overall catheter performance
- Double-braid wire for enhanced torque, strength, and kink resistance
- InSlide™ polymer additive for superior lubricity and enhanced deliverability1
- Soft, atraumatic, radiopaque tip for excellent visibility
- Large lumen for high flow of contrast media to increase visualization2
Features and Benefits
Enhanced deliverability:
§ InSlide™ polymer additive for superior lubricity to
reduce friction and enhance deliverability2
Precise manipulation:
§ Unique, ergonomic, winged hub design for easy
manipulation
§ Double-braid wire applied from hub to tip enhances
strength, torque and kink resistance
Excellent image quality:
§ Soft, atraumatic radiopaque tip allows for increased
visibility
§ Large lumen enables high flow of contrast media to
increase visualisation
Broad catheter portfolio:
§ Optimised for both radial and femoral procedures
§ Wide range of shapes and sizes
§ Includes expanded offerings of long (125 cm) lengthsMarket-leading technology improves
performance where it matters:
§ Superior torque and lubricity for precise
manipulation and enhanced deliverability3
§ Optimised for both radial and femoral procedures4
§ Wide range of shapes and sizesOrdering Information
Curve Style
Curve
Size
Length
(cm) Product Code Sideholes
Judkins Left JL 3.5 100 DXT5JL35 0
JL 4.0 100 DXT5JL40 0
JL 4.0 125 DXT5JL40X 0
JL 4.5 100 DXT5JL45 0
JL 4.5 125 DXT5JL45X 0
JL 5.0 100 DXT5JL50 0
JL 5.0 125 DXT5JL50X 0
JL 6.0 100 DXT5JL60 0
Judkins Right JR 3.5 100 DXT5JR35 0
JR 3.5 125 DXT5JR35X 0
JR 4.0 100 DXT5JR40 0
JR 4.0 125 DXT5JR40X 0
JR 4.0 ST 100 DXT5JR4ST 0
JR 4.0 MOD 100 DXT5JR4M 0
JR 5.0 100 DXT5JR50 0
JR 5.0 125 DXT5JR50X 0
JR 6.0 100 DXT5JR60 0
Amplatz Left AL 1.0 100 DXT5AL10 0
AL 2.0 100 DXT5AL20 0
AL 3.0 100 DXT5AL30 0
Amplatz Right AR 1.0 100 DXT5AR10 0
AR 2.0 100 DXT5AR20 0
AR MOD 100 DXT5ARM 0
Multipurpose MPA 100 DXT5MPA 0
MPA 110 DXT5MPAA 0
MPA 100 DXT5MPASH 2
MPA 110 DXT5MPASHA 2
MPB 100 DXT5MPB 0
MPB 110 DXT5MPBA 0
MPB 100 DXT5MPBSH 2
MPB 110 DXT5MPBSHA 2
Specialty NOTO 100 DXT5NOTO 0
3DRC 100 DXT53DRC 0
RCB 100 DXT5RCB 0
LCB 100 DXT5LCB 0
IMA 100 DXT5IMA 0
Pigtails PIG STR 110 DXT5PIGSTA 6
PIG STR 125 DXT5PIGSTX 6
PIG 145 110 DXT5PIG45A 6
PIG 155 110 DXT5PIG55A 6 -
-
Elastic Stay Hooks
Elastic Stay Hooks
From delicate tissues to substantial anatomical structures, the Lone Star Elastic Stays provide the retraction necessary to achieve and maintain optimal visualization throughout a variety of procedures. Available in both sharp and blunt options, the Elastic Stays fit securely within the slots of the Lone Star Retractors ensuring reproducible, reliable results case after case.
Parts & Accessories
3311-1G 5 mm sharp hook (50/Case) 3311-8G 5 mm sharp hook (8/Pack) 3314-1G 3 mm sharp hook (50/Case) 3314-8G 3 mm sharp hook (8/Pack) 3325-4G 5 mm two-finger (4/Pack) 3327-4G 7 mm two-finger (4/Pack) 3329-4G 9 mm two-finger (4/Pack) 3330-4G 13 mm x 16 mm solid blade (4/Pack) 3331-4G 13 mm Spira blunt hook (4/Pack) 3332-4G 14 mm x 16 mm two-finger (4/Pack) 3333-4G 16 mm Spira two-finger (4/Pack) 3334-4G 20 mm x 16 mm four-finger (4/Pack) 3335-4G 20 mm Spira three-finger (4/Pack) 3338-4G 6.5 mm x 16 mm solid blade (4/Pack) 3350-1G 12 mm blunt hook (50/Case) 3350-8G 12 mm blunt hook (8/Pack) 3350L-4G 12 mm blunt hook, elongated lead (4/Pack) 3384-4 12 mm two-finger blunt hook (4/Pack) 3550-1G 12 mm blunt hook, dual lead (50/Case) 3316-1G 5 mm blunt hook (50/case) 3316-8G 5 mm blunt hook (8/Pack) 3205-8G 5 mm semi-blunt PRO-Stay (8/Pack) -
ELIMINATE™ – ASPIRATION CATHETER
Description
A fast and simple solution for effective thrombus aspiration
Eliminate is an innovative aspiration catheter designed to offer a balance between crossing performance, kink resistance and thrombus-aspiration capability.
Aspiration catheter. The deliverable solution for thrombus removal. Guide catheter compatibility. EliminateTM. 6Fr. (I.D. ≥ 0.70 inch / 1.78 mm).
Eliminate™ is an aspiration catheter intended for removing fresh, soft emboli and thrombi from vessels in the coronary and peripheral vasculature.
Initial experience with a mechanical aspiration catheter for thrombus removal during … Catalog. Number. Description. Distal OD (in). Total Length.
Characteristics
Dedicated tip design with radiopaque marker
- Excellent crossability1 while providing atraumatic and effective aspiration
- Ensures reliable fluoroscopic visibility
Braided shaft with hydrophilic coating and pre-loaded stiffening stylet2
- Excellent pushability and kink resistance1
- Easy navigation through tortuous anatomies
Large aspiration lumen
- Constant, high-performance aspiration throughout the procedure
- Choice of three sizes (6 Fr, 7 Fr and 8 Fr guide catheter compatibility) for different coronary and peripheral applications
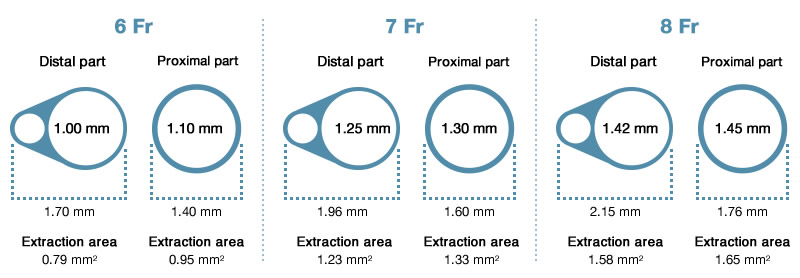
Guide Catheter Compatibility
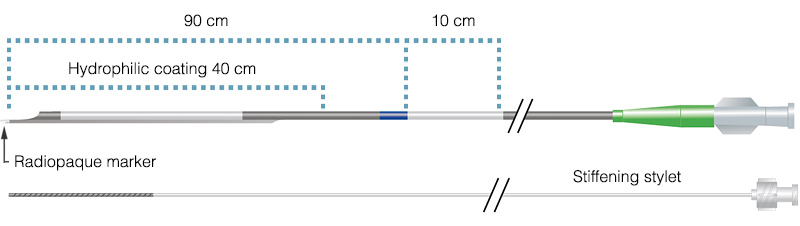
General specifications
Usable length 140 cm Distal tip hole length 4 mm for 6 and 7 Fr, 7 mm for 8 Fr Rapid exchange segment 23 cm Radiopaque marker 1 mm located at 4 mm from the tip Positioning marker 10 cm = single white mark located at 90 cm from distal tip Guidewire compatibility Maximum diameter 0.014″ (0.36 mm) Coating Hydrophilic Item specifications
Guide catheter compatibility Guide sheath compatibility Item reference 6 Fr I.D. ≥ 0.070″ / 1.78 mm — EG1602 7 Fr I.D. ≥ 0.080″ / 2.03 mm — EG1652 8 Fr I.D. ≥ 0.086″ / 2.18 mm 6 Fr EG1401 Please quote above item reference codes when placing an order.
References
1 Onuma et al.; Eur Heart J. 2013 Apr;34(14):1050-60. doi: 10.1093/eurheartj/ehs456. Epub 2013 Feb 8.
2 Only the 6 Fr and 7 Fr options are pre-loaded. -
ELISIO-15M
ELISIO is a synthetic hollow-fiber dialyzer with a unique polynephron membrane that delivers outstanding performance, excellent biocompatibility, and low thrombogenicity. Dialyzer used in Hemo Dialysis
-
Emboshield NAV6 Embolic Protection System
Emboshield NAV6 Embolic
Indicated for Carotids and Lower Extremities
The Emboshield NAV6™ Embolic Protection System, which includes BareWire™ Filter Delivery Wires, allows the guide wire to rotate and advance freely, independent of the Emboshield NAV6™ filter.*
The Emboshield NAV6™ EPS is indicated for use as a guide wire and embolic protection system to contain and remove embolic material (thrombus / debris) while performing angioplasty and stenting procedures in carotid arteries and while performing atherectomy, during standalone procedures or together with PTA and/or stenting, in lower extremity arteries. The diameter of the artery at the site of the Filtration Element placement should be between 2.5 and 7.0 mm.
EMBOLIC PROTECTION DEVICE FOR BOTH LOWER EXTREMITY AND CAROTID ARTERIES
Captures Effectively to Minimize Downstream Complications1-3
- Centered wire design prevents bias against the vessel wall for effective debris capture.*
- Circumferential nitinol frame maintains optimal wall apposition, even on a bend.*
- Platinum-tungsten frame coils provide excellent visibility.*
- Two sizes allow for easy selection and minimal lab inventory.
Limits Filter Movement and Maintains Wire Access Through Innovative BareWire™ Design*
- The unique BareWire™ technology allows the wire to rotate and advance freely, independently of the filter.
- The filter is designed to stay in place during device delivery.
- Continued wire access, after filter is fully retracted, allows for easy delivery of additional therapy.
Navigates Skillfully Through Carotid and Lower Extremity Vasculature*
- Various types of BareWires™—distal access, workhorse, and support—are designed for various carotid anatomies.
- They promote navigational success through torturous anatomy and challenging arches.
Is Compatible with Various Atherectomy Options4
- Emboshield NAV6™ EPS and BareWire™ are compatible with a variety of atherectomy types.
- BareWire™ is available in 190 cm and 315 cm lengths.
In carotid procedures, the Emboshield NAV6™ EPS is used during stenting with the RX Acculink™ Carotid Stent System and the Xact™ Carotid Stent System.
* Data on file at Abbott.
References
- Bioangiu et al. Comparative analysis of retrieved particulate debris after superficial femoral atherectomy using three different atherectomy methods. EuroIntervention, May 2012.
- Bioangiu et al. Analysis of Retrieved Particulate Debris after Superficial Femoral Artery (SFA) Atherectomy Using the Pathway Jetstream G3 device. CCI, May 2011, 77(2) p. S57.
- Mendes et al. Clinical significance of embolic events in patients undergoing endovascular femoropopliteal interventions with or without embolic protection devices. JVS, February 2014, 59(2), 359-367.
- Philips Turbo-Elite, Medtronic TurboHawk PPES, and Boston Scientific Jetstream XC and SC. Test(s) performed by and data on file at Abbott.
IMPORTANT SAFETY INFORMATION
 Emboshield NAV6 Embolic Protection System
Emboshield NAV6 Embolic Protection SystemIndications
The Emboshield NAV6 Embolic Protection System is indicated for use as a guide wire and embolic protection system to contain and remove embolic material (thrombus / debris) while performing angioplasty and stenting procedures in carotid arteries and while performing atherectomy, during standalone procedures or together with PTA and/or stenting, in lower extremity arteries. The diameter of the artery at the site of the Filtration Element placement should be between 2.5 and 7.0 mm.
Contraindications
The Emboshield NAV Embolic Protection System is contraindicated for use in
- Patients in whom anticoagulant and / or antiplatelet therapy is contraindicated.
- Patients with severe vascular tortuosity or anatomy that would preclude the safe introduction of the Guiding Catheter / Introducer Sheath, Embolic Protection System.
- Patients with a known allergy or hypersensitivity to device materials (Nitinol, Nickel, Titanium) or contrast medium, who cannot be adequately premedicated.
- Patients with uncorrected bleeding disorders.
- Lesions in the ostium of the common carotid artery.
- Inability to cross the lesion with the BareWire™ Filter Delivery Wire.
- Diffusely diseased vessels where there is no disease-free section in which to deploy the Filtration Element
- Insufficient straight section of vessel distal to the lesion to permit Filtration Element deployment.
-
EMPIRA PTCA BALLOON
Product Description
EMPIRA™ PTCA Balloons – CordisEMPIRA™ Pre Dilatation and Post Dilatation PTCA Balloons catheters helps you reach and treat a wide range of lesions with exceptional crossability
EMPIRA™ NC Balloon Catheter delivers crossability & best-in-class controlled growth to a wide range of lesions. EMPIRA™ NC RX PTCA
The Cordis PTCA Balloon Portfolio includes the EMPIRA™ Balloon, which helps to treat a wide range of lesions with exceptional crossability and redressability.The Cordis PTCA Balloon Portfolio includes the EMPIRA™ Balloon, which helps to treat a wide range of lesions with exceptional crossability and redressability.
-
ENDO CLIP 10MM
The Endo Clip 10 mm applier contains 20 titanium clips in the ML and M sizes, and 15 titanium clips in the L size. The applier is designed for introduction and use through all appropriately sized trocar sleeves, or larger sized trocar sleeves with the use of a converter. -
Endo Gia Stapler
Endo Gia Stapler system enables all surgeons to operate with confidence to handle the broadest range of tissues and applications with outstanding clinical performance
- Accurate staple formation
- Reduces the risks for cross-contamination
- Less stress on the tissue
- Improved maneuverability
- State-of-the-art design
-
Endosee Advance System
Endosee Advance System
Introducing the Endosee Advance system! The Endosee Advance system is an all-in-one, direct visualization system for diagnostic and therapeutic procedures. Advance patient care and your practice
- Instant endometrial imaging guided by direct visualization1
- Efficient workflow with in-office exams that better prepare for and reduce OR visits2,3
- Low-cost investment that is coded and reimbursed as hysteroscopy3
Features to help elevate your in-office procedures:
- Your exam is guided by clear color visualization
- Reusable display module
- Sterile, single-use cannula designed for ease of insertion
- Channel for fluid infusion
- Still image or video capture of every exam
- See the picture you just took
- Transfer exam data to your computer via USB cable
1 Goldstein SR, Anderson TL. Endometrial Evaluation: Are you still relying on a blind biopsy? OBG Mgmt Supp. 2017;10:S1-S4.
2 Isaacson K. Office Hysteroscopy: a valuable but under-utilized technique. Curr Opin Obstet Gynecol. 2002;14:381-385. 3 Moawad N, Santamaria E, Johnson M, Shuster J. Cost effectiveness of office hysteroscopy for abnormal bleeding. JSLS. 2014;18:1-5.
Parts & Accessories
ES9000 Endosee Advance Display Module (1 unit) ESPX5 Endosee Cannula, compatible with ES9000 only (5 per Box) ES-TRAY Endosee System Convenience Kit (5 per Box) ES-BPSY Biopsy forceps ES–LNGR Spoon forceps, long serrated ES-FBGR Foreign Body Grasper, fenestrated ES-SCIS Scissors, single action ES-DISP-AGR Disposable Alligator Grasper Forceps, 5 Fr, 460 mm (5/pack) ES-DISP-SCS Disposable Single Action Scissors, 5 Fr, 460 mm ES-DISP-BPY Disposable Biopsy Forceps, 5 Fr, 460 mm 53604 Cystometry Tube Sets (10 Sets/Box) ESSYR 50 mL Syringe (sterile 40 per case) ESTUB Standard Bore Extension Tubing 30 in. (sterile 100 per case) -
Endovascular Snare
EN Snare Endovascular Snare
System
NAVIGATE. CAPTURE. SUCCEED.
The EN Snare Endovascular Snare System is designed with three interlaced loops to retrieve and manipulate foreign objects in the body. The EN Snare can be used to retrieve inferior vena cava filters, reposition indwelling venous catheters, fibrin sheath stripping, or to assist in central venal access venipuncture. Super elastic Nitinol provides flexibility, kink resistance, and torque control. The EN Snare is designed to rotate and expand for excellent coverage and retrieval within a variety of vessel sizes. Platinum strands are incorporated into the EN Snare loops for excellent visualization under fluoroscopy.
ORDER INFORMATION
CATALOG NUMBER EXPANDED FAMILY NAME1 EXPANDED FAMILY NAME2 SNARE LENGTH (CM) SNARE LENGTH (INCHES) SNARE WORKING DIAMETER (MM) SNARE WORKING DIAMETER (INCHES) SNARE COLLAPSED DIAMETER EN-C03150 EN Snare® Endovascular Snare EN-C06100 EN Snare® Endovascular Snare EN-C07100 EN Snare® Endovascular Snare EN1003004 EN Snare® Endovascular Snare System 175 cm (69″) 2 mm – 4 mm (0.08″ – 0.16″) 0.028″ (0.71 mm) EN1003008 EN Snare® Endovascular Snare System 175 cm (69″) 4 mm – 8 mm (0.16″ – 0.32″) 0.028″ (0.71 mm) EN2006010 EN Snare® Endovascular Snare System 120 cm (47″) 6 mm – 10 mm (0.23″ – 0.39″) 0.045″ (1.14 mm) EN2006015 EN Snare® Endovascular Snare System 120 cm (47″) 9 mm – 15 mm (0.35″ – 0.59″) 0.055″ (1.40 mm) EN2006020 EN Snare® Endovascular Snare System 120 cm (47″) 12 mm – 20 mm (0.47″ – 0.79″) 0.055″ (1.40 mm) EN2007030 EN Snare® Endovascular Snare System 120 cm (47″) 18 mm – 30 mm (0.71″ – 1.18″) 0.055″ (1.40 mm) EN2007045 EN Snare® Endovascular Snare System 120 cm (47″) 27 mm – 45 mm (1.06″ – 1.77″) 0.055″ (1.40 mm) -
EUPHORA SEMICOMPLIANT
EUPHORA SEMICOMPLIANT
OVERVIEW
Advancing Innovation through Partnership
To develop the Euphora™ low-profile semicompliant balloon dilatation catheter, we partnered with nearly 2,000 interventional cardiologists and cath lab professionals. The result is:
- Superior deliverability1
- Superb Kissing Balloons Technique (KBT) performance2
- The lowest crossing profile compared to leading competitors3
PRODUCT DETAILS
BALLOON LENGTHS 6, 10, 12, AND 15 MM
Columns1.50 EUP1506X EUP1510X EUP1512X EUP1515X 2.00 EUP2006X EUP2010X EUP2012X EUP2015X 2.25 EUP22506X EUP22510X EUP22512X EUP22515X 2.50 EUP2506X EUP2510X EUP2512X EUP2515X 2.75 EUP27506X EUP27510X EUP27512X EUP27515X 3.00 EUP3006X EUP3010X EUP3012X EUP3015X 3.25 EUP32506X EUP32510X EUP32512X EUP32515X 3.50 EUP3506X EUP3510X EUP3512X EUP3515X 3.75 EUP37506X EUP37510X EUP37512X EUP37515X 4.00 EUP4006X EUP4010X EUP4012X EUP4015X BALLOON LENGTHS 20, 25, AND 30 MM
Columns1.50 EUP1520X — — 2.00 EUP2020X EUP2025X EUP2030X 2.25 EUP22520X EUP22525X — 2.50 EUP2520X EUP2525X EUP2530X 2.75 EUP27520X EUP27525X — 3.00 EUP3020X EUP3025X EUP3030X 3.25 EUP32520X EUP32525X — 3.50 EUP3520X EUP3525X EUP3530X 3.75 EUP37520X EUP37525X — 4.00 EUP4020X EUP4025X EUP4030X -
EUPHORA SEMICOMPLIANT BALLOON CATHETER
Euphora™ semicompliant balloon
Balloon Dilatation Catheter
Euphora™ semicompliant balloon dilatation catheter complements your expertise and provides uncompromising performance in the moment you need it most.
OVERVIEW
Advancing Innovation through Partnership
To develop the Euphora™ low-profile semicompliant balloon dilatation catheter, we partnered with nearly 2,000 interventional cardiologists and cath lab professionals. The result is:
- Superior deliverability1
- Superb Kissing Balloons Technique (KBT) performance2
- The lowest crossing profile compared to leading competitors3
PRODUCT DETAILS
What Makes the Euphora Balloon an Excellent Choice?
ORDERING INFORMATION
BALLOON LENGTHS 6, 10, 12, AND 15 MM
1.50 EUP1506X EUP1510X EUP1512X EUP1515X 2.00 EUP2006X EUP2010X EUP2012X EUP2015X 2.25 EUP22506X EUP22510X EUP22512X EUP22515X 2.50 EUP2506X EUP2510X EUP2512X EUP2515X 2.75 EUP27506X EUP27510X EUP27512X EUP27515X 3.00 EUP3006X EUP3010X EUP3012X EUP3015X 3.25 EUP32506X EUP32510X EUP32512X EUP32515X 3.50 EUP3506X EUP3510X EUP3512X EUP3515X 3.75 EUP37506X EUP37510X EUP37512X EUP37515X 4.00 EUP4006X EUP4010X EUP4012X EUP4015X BALLOON LENGTHS 20, 25, AND 30 MM
1.50 EUP1520X — — 2.00 EUP2020X EUP2025X EUP2030X 2.25 EUP22520X EUP22525X — 2.50 EUP2520X EUP2525X EUP2530X 2.75 EUP27520X EUP27525X — 3.00 EUP3020X EUP3025X EUP3030X 3.25 EUP32520X EUP32525X — 3.50 EUP3520X EUP3525X EUP3530X 3.75 EUP37520X EUP37525X — 4.00 EUP4020X EUP4025X EUP4030X -
Everest30 INFLATION DEVICE
Everest 30
Disposable Inflation Device
OVERVIEW
Easy-to-use Rapid Inflation/Deflation Device
A simple solution for coronary balloon inflation. The Everest disposable inflation device features a quick trigger release and ergonomic pistol grip for ease of use. Plus, it’s available in your choice of 20 atm or 30 atm pressure gauge, and as a standalone or in a packaged kit.
PRODUCT DETAILS
The Everest Device Features:
- Quick trigger release so you can release pressure immediately, even at higher pressures
- Sure-grip torque knob with a ridged design for “no slip” torque control and precise pressure adjustment
- Unique ergonomic pistol grip with centered trigger that fits comfortably in either hand
- Luminescent dial face that provides accurate readability in normal and low-light conditions
EVEREST DISPOSABLE INFLATION DEVICE ORDERING INFORMATION
Disposable Inflation Device@ AC3200
Survival Kit Inflation Device@ AC305P
Everest20 inflation device
and 3-way stopcockAC2200 1 each Everest30 inflation device
and 3-way stopcockAC3200 1 each Everest20 Survival Kit: - Everest20 inflation device
- 3-way stopcock
- Piton Y-Adapter
- Metal guidewire insertion tool
- Torque handle
AC2205P 1 each Everest30 Survival Kit: - Everest30 inflation device
- 3-way stopcock
- Piton Y-Adapter
- Metal guidewire insertion tool
- Torque handle
AC3205P 1 each ACCESSORIES ORDERING INFORMATION
Metal guidewire insertion tool 006073 5 each Torque handle AC4006 5 each Piton™ Y-Adapter with metal guidewire insertion tool AC4001M 5 each Piton™ Tri-Adapter with metal guidewire insertion tool AC4002M 5 each Piton™ Y-Adapter with metal guidewire insertion tool and torque handle AC4003M 5 each -
EXPORT ADVANCE ASPIRATION CATHETER
EXPORT ADVANCE ASPIRATION CATHETER
Export Advance
Aspiration Catheter
ADVANCING PERFORMANCE
The Export Advance™ aspiration catheter delivers consistent, high-performing aspiration power when it matters most — restoring flow and protecting patients.
PRODUCT DETAILS†
Superior Deliverability1
Full-Wall Technology provides variable levels of stiffness without joints, for optimal kink resistance and catheter performance.
Dependable Delivery to the Target Site:
- Preloaded stylet enhances shaft stiffness during catheter delivery, boosting kink resistance, trackability, and pushability2
- Buddy wire compatibility allows for extra support, when needed.†
High-performing Aspiration Power:
- Large extraction lumen (0.044″ proximal; 0.043″ distal) increases aspiration power.‡
- Optimised hub geometry improves flow.‡
- Soft, short, forward-facing tip design permits excellent particle capture.†
ORDERING INFORMATION
ADVANCE 6 F min. guide I.D. 0.070″ 0.014 140 - ™Third-party brands are trademarks of their respective owners. All other brands are trademarks of a Medtronic company.
- Based on bench test data, 2013. Bench test data may not be indicative of clinical performance.
- Based on bench test data vs. Export™ AP catheter.
- Bench testing vs. Terumo Eliminate™* and Eliminate™* 3 aspiration catheters, Vascular Solutions Pronto®* LP, Pronto®* V3 and Pronto®* V4 aspiration catheters, Merit®* ASAP aspiration catheter, Kaneka Thrombuster III GR* aspiration catheter, Spectranetics QuickCat™* aspiration catheter, IHT Polska Hunter®* aspiration catheter, Medrad Possis™* Fetch™* 2 aspiration catheter, and Hexacath Recover* aspiration catheter. 10093906DOC, 2013. Bench test data may not be indicative of clinical performance.
- Based on animal studies compared with Export™ AP aspiration catheter without stylet.
-
-
FIELDER® FC
FIELDER® FC
Greater control in challenging tortuous vessels/highly stenosed lesions. Polymer jacket provides advanced lubricity for superior torque.

TIP LOAD 
TIP RADIOPACITY 3 cm COATING POLYMER JACKET & HYDROPHILIC*
Hydrophilic coating over polymer jacket: 20cmTIP SHAPE 

Ordering Information
PRODUCT CATALOG NO. TIP SHAPE LENGTH (CM) DIAMETER (INCH(MM)) RADIOPAQUE SEGMENT (CM) SPRING COIL LENGTH (CM) COATING FIELDER FC 180cm, Straight AGP140001 Straight 180 0.014 (0.36) 3 11 POLYMER JACKET & HYDROPHILIC*
Hydrophilic coating over polymer jacket: 20cmFIELDER FC 180cm, J Shape AGP140001J J Shape 180 0.014 (0.36) 3 11 POLYMER JACKET & HYDROPHILIC*
Hydrophilic coating over polymer jacket: 20cmFIELDER FC 300cm, Straight AGP140301 Straight 300 0.014 (0.36) 3 11 POLYMER JACKET & HYDROPHILIC*
Hydrophilic coating over polymer jacket: 20cmIndications for Use
ASAHI PTCA Guide Wires are intended to facilitate the placement of balloon dilation catheters during percutaneous transluminal coronary angioplasty (PTCA) and percutaneous transluminal angioplasty (PTA). The ASAHI PTCA Guide Wires are not to be used in the neurovasculature.
-
FIELDER® XT
Chronic Total Occlusion Guide Wires

FIELDER® XT
Good choice for narrow channel crossing due to the tapered flexible tip combined with the highly lubricious coating. Chronic Total Occlusion indicated.

TIP LOAD 
TIP RADIOPACITY 16 cm COATING POLYMER JACKET & HYDROPHILIC*
Hydrophilic coating over polymer jacket: 16cmTIP SHAPE 
Ordering Information
PRODUCT CATALOG NO. TIP SHAPE LENGTH (CM) DIAMETER (INCH(MM)) RADIOPAQUE SEGMENT (CM) SPRING COIL LENGTH (CM) COATING FIELDER XT 190cm AGP140002 Straight 190 0.009/0.014 (0.23/0.36) 16 16 POLYMER JACKET & HYDROPHILIC*
Hydrophilic coating over polymer jacket: 16cmFIELDER XT 300cm AGP140302 Straight 300 0.009/0.014 (0.23/0.36) 16 16 POLYMER JACKET & HYDROPHILIC* Indications for Use
ASAHI PTCA Guide Wires are intended to facilitate the placement of balloon dilation catheters during percutaneous transluminal coronary angioplasty (PTCA) and percutaneous transluminal angioplasty (PTA). The ASAHI PTCA Guide Wires are not to be used in the neurovasculature.
-
FINECROSS Microcatheter
FINECROSS® MG Coronary Micro-Guide Catheter
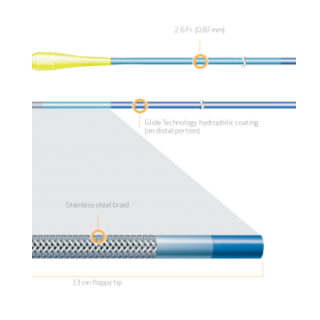
Product Code Catheter Length Distal Hydrophilic Coating Distal Outer Diameter Proximal Outer Diameter Distal Inner Diameter Proximal Inner Diameter Recommended Guidewire Size 35-1430 130cm 70cm 1.8Fr. (0.60mm) 2.6 Fr. (0.87mm) 0.018″ (0.45mm) 0.021″ (0.55mm) 0.014″ (0.36mm) 35-1450 150cm 90cm 1.8Fr. (0.60mm) 2.6 Fr. (0.87mm) 0.018″ (0.45mm) 0.021″ (0.55mm) 0.014″ (0.36mm) PRODUCT OVERVIEW
Coronary microguide catheter for integration of optimal guidewire support, superior trackability and crossability.1,2

TAPERED STAINLESS STEEL BRAID CONSTRUCTION FOR OPTIMAL GUIDEWIRE SUPPORT
FINECROSS MG stainless steel braid construction is designed to provide strength, responsiveness and support for improved pushability to access and cross complex lesions.
13cm FLOPPY DISTAL SEGMENT FOR SUPERIOR TRACKABILITY1
The distal 13 cm is ultra flexible for improved trackability around tight bends and tortuous anatomy.The floppy distal segment is designed to be atraumatic and provide an optimal balance between trackability and safety while navigating through the tortuous anatomy.1
TAPERED OUTER DIAMETER FOR SUPERIOR CROSSABILITY1,2
The outer diameter of the stainless steel shaft tapers from a proximal 2.6Fr. to a distal 1.8Fr. designed for improved crossability and guidewire handling.1,2
DOCUMENTS
-
FINECROSS® MICRO-GUIDE CATHETHER
PRODUCT OVERVIEW
Coronary micro guide catheter for integration of optimal guidewire support, superior trackability and crossability.FINECROSS® MG Coronary Micro-Guide Catheter is designed to provide the ultimate balance of properties for access and crossing of complex lesions.
Terumo has been providing TRI-related products since the 80s. Browse our products today. Radial Edge Solutions reduce bleeding rates, in-hospital mortality, and related costs. Designed & Made in Japan. Fine Hydrophilic Coating. Pioneer in Radial Access.
Introduction
Coronary lesions often exhibit certain features posed as challenging while performing a percutaneous coronary intervention (PCI) viz. thrombus, bifurcation, ostial lesion, calcification, and chronic total occlusions (CTOs) (1). The use of microcatheters as a coronary interventional tool assists in performing invasive endovascular procedures by serving a broad range of functions in these complex vessel anatomies.
The objective of this article is to formulate and deliberate on the recommendations for the selection of microcatheters and their utility in coronary interventional procedures. The discussion included broadly the following two topics:
• Understanding the significance of a microcatheter in the cardiac catheterization lab—which included a discussion on the types of microcatheter available in the Indian market and their significance in PCI procedures.
• Criteria for the selection of microcatheters—which included a discussion on the selection and preference of one microcatheter over another based on lesion/anatomy-specific criteria or hardware characteristics.
TAPERED STAINLESS STEEL BRAID CONSTRUCTION FOR OPTIMAL GUIDEWIRE SUPPORT
FINECROSS MG stainless steel braid construction is designed to provide strength, responsiveness and support for improved pushability to access and cross complex lesions.
13cm FLOPPY DISTAL SEGMENT FOR SUPERIOR TRACKABILITY
The distal 13 cm is ultra flexible for improved trackability around tight bends and tortuous anatomy. The floppy distal segment is designed to be atraumatic and provide an optimal balance between trackability and safety while navigating through the tortuous anatomy.
TAPERED OUTER DIAMETER FOR SUPERIOR CROSSABILITY
The outer diameter of the stainless steel shaft tapers from a proximal 2.6Fr. to a distal 1.8Fr. designed for improved crossability and guidewire handling.
FINECROSS MG Product SKUs
Product Code Catheter Length Distal Hydrophilic Coating Distal Outer Diameter Proximal Outer Diameter Distal Inner Diameter Proximal Inner Diameter Recommended Guidewire Size 35-1430 130cm 70cm 1.8Fr. (0.60mm) 2.6 Fr. (0.87mm) 0.018″ (0.45mm) 0.021″ (0.55mm) 0.014″ (0.36mm) 35-1450 150cm 90cm 1.8Fr. (0.60mm) 2.6 Fr. (0.87mm) 0.018″ (0.45mm) 0.021″ (0.55mm) 0.014″ (0.36mm Structural Properties/Characteristics of Microcatheters
As per the experts, multiple microcatheter features impact its selection viz. tip profile, shaft profile, tip and shaft flexibility, inner lumen diameter, radiopacity of the device, and microcatheter tube structure a.
Conclusion
Thus, microcatheter selection and operating techniques are vital to a successful treatment plan, which should be based on the recommendations derived for clinical expertise and operator efficiency. For next-generation microcatheters, an increase in support without compromising on flexibility and lowering of the tip profile in comparison to the ones currently present in the market is needed.
-
FLOPPY II
FLOPPY II GUIDE WIRE
HI-TORQUE FLOPPY II is a light support wire known for its soft, gentle tip. The HI-TORQUE FLOPPY II has light support while the FLOPPY II ES has extra support for device delivery.
HI-TORQUE FLOPPY II Guide Wire
Tip load: 0.4 g
Radiopaque length: 2 cm
Outside diameter: 0.014″
Tip Outside diameter: 0.014″
Coating: Hydrophilic or Hydrophobic
Tip style: Shaping Ribbon
Polymer cover: none
Core Material: Stainless SteelHI-TORQUE FLOPPY II ES Guide Wire
Tip load: 0.6 g
Radiopaque length: 2 cm
Outside diameter: 0.014″
Tip Outside diameter: 0.014″
Coating: Hydrophilic or Hydrophobic
Tip style: Shaping Ribbon
Polymer cover: noneCore Material: Stainless Steel
HI-TORQUE FLOPPY II™ Family of Guide Wires
PRODUCT PART NUMBER TIP RADIOPACITY TIP SHAPE DISTAL COATINGS WIRE LENGTH HI-TORQUE FLOPPY II™ 22339H 2 cm Straight Hydrophilic 190 cm HI-TORQUE FLOPPY II™ 22339M 2 cm Straight Hydrophobic 190 cm HI-TORQUE FLOPPY II™ 22339MJ 2 cm J Hydrophobic 190 cm HI-TORQUE FLOPPY II™ 22359M 2 cm Straight Hydrophobic 300 cm HI-TORQUE FLOPPY II™ 22359MJ 2 cm J Hydrophobic 300 cm HI-TORQUE FLOPPY II™ 22339M-903 30 cm Straight Hydrophobic 190 cm HI-TORQUE FLOPPY II™ ES 22299H 2 cm Straight Hydrophilic 190 cm HI-TORQUE FLOPPY II™ ES 22299M 2 cm Straight Hydrophobic 190 cm HI-TORQUE FLOPPY II™ ES 22299M-901 30 cm Straight Hydrophobic 190 cm HI-TORQUE FLOPPY II™ ES 22359M-901 2 cm Straight Hydrophobic 300 cm -
FORGATY CATHETER
Fogarty arterial embolectomy catheter
Removal of fresh, soft emboli and thrombi from vessels in the arteries of the non-central circulatory system
- Hand-tied using a recessed winding technique to secure balloon to catheter
- Balloon exhibits a symmetry that exerts uniform contact with vessel walls for even pressure and
precise traction - Rounded tip promotes easy insertion for reduced trauma
Models
Fogarty arterial embolectomy catheters (5 pack)
Model Length (cm) Catheter Size (F) Inflated Balloon Diameter (mm) Max. Size of Deflated Balloon (F) Max. Gas Capacity (ml) Max. Liquid Capacity (ml) Max. Recommended Pull Force on Inflated Balloon (lbs.) Compatible Guidewire (in.) 120602FP 60 2 4 3.4 0.2 – 0.5 – 120403FP 40 3 5 4.3 0.6 0.20 0.7 – 120403FSP 40 3 5 4.3 0.6 0.20 0.7 – 120803FP 80 3 5 4.3 0.6 0.20 0.7 – 120803FSP 80 3 5 4.3 0.6 0.20 0.7 – 120404FP 40 4 9 5.0 1.7 0.75 1.5 – 120804FP 80 4 9 5.0 1.7 0.75 1.5 – 120805FP 80 5 11 6.0 3.0 1.50 2.0 – 120806FP 80 6 13 7.0 4.5 2.00 2.5 – 120807FP 80 7 14 8.0 5.0 2.50 3.5 – Accessories
Edwards Fogarty catheter rack
- This rack is intended to be wall-mounted
- After wall-mounting it can be used to hold Fogarty catheters
- Model number CV6000
-
Fresenius F6 HPS Dialyzer, For Haemodialysis
F-Series
Not all polysulfones are like Fresenius
Polysulfone
- Proven quality and efficiency of Fresenius Polysulfone
- The original benchmark for biocompatibility
- INLINE steam sterilized
- Available in both low-flux and high-flux ranges

Key features
Fresenius Polysulfone® — A synonym for biocompatibility
- Adverse interactions between blood and material arising during the dialysis treatment have been related to a variety of clinical symptoms
- It has become increasingly evident that not all dialysis membranes are the same with respect to their ability to activate blood cell components1
- Ever since its introduction, Fresenius Polysulfone® has been recognized for its biocompatibility and was an established benchmark for all membrane improvements and developments 2,3,4,5
Safety during each and every treatment
- All Fresenius Medical Care dialyzers provide a high degree of safety during each and every treatment
- The high endotoxin retention capabilities of the Fresenius Polysulfone® are attributed to the chemical composition and structure of the membrane
Enhanced design for maximum efficiency
- The special undulating structure of the hollow fibers delivers an excellent performance profile
- Spacing between the wavy fiber structures keeps the individual fibers apart, preventing dialyzate channeling and facilitating a uniform flow of dialyzate within the fiber bundle
- The consistent dialyzate flow around each fiber ensures every single performs at maximum efficiency, resulting in enhanced solute transport
Technology
INLINE steam sterilization – purity enhanced
No chemical residuals. Low rinsing volumes. Lower costs.
INLINE steam sterilization – how it works
- Both the blood and the dialysate compartment of the dialyzers are rinsed continuously with steam at a temperature at or above 121°C for a minimum of 15 minutes. Rinsing with hot steam and without chemicals results in extremely low levels of residuals in the dialyzer
- The dialyzer is rinsed with sterile water
- Every dialyzer is tested for fiber integrity using a bubble-point test
- The dialyzers are dried with warm, sterile air
- Finally, after drying the blood inlet and outlet ports are closed
INLINE steam sterilization – the benefits
- Highly pure, sterile and pyrogen-free dialyzers without any potentially harmful residuals from sterilization
- Biocompatibility of membranes remains unaffected from sterilization
- Optimized use of resources due to low rinsing volumes: only 500 mL is required
- Dry dialyzers with minimized risk of contamination due to microbial growth
Fibre integrity testing
- All dialyzers have to pass the bubble point test as part of the INLINE steam sterilization process
- Sterile air is pressed into the dialyzate compartment while the blood compartment contains sterile water
- If any leakages were present in the membrane, air would pass through the membrane and create bubbles
- Dialyzers failing the integrity test are discarded
- This integrity test minimizes the risk of fibre ruptures and the risk of blood leakages
Quality from start to finish
- Fresenius Polysulfone® dialyzers are produced to meet international safety regulations
- Fresenius Medical Care’s core competency is the complete production from membranes to the finished dialyzers, providing the highest quality during every step of production
Performance data
Low Flux Dialysers – High Performance Steam (HPS)
Low Flux Dialysers – High Performance Steam (HPS) F4 HPS F5 HPS F6 HPS F7 HPS F8 HPS F10 HPS Ultrafiltration coefficient (ml/h x mmHg) 8 10 13 16 18 21 Clearance: QB: (200ml/min) Urea 170 179 186 188 Creatinine 149 162 173 175 Phosphate 123 139 148 155 Vitamin B12 75 84 92 102 Clearance: QB: (300ml/min) Urea 227 243 247 252 259 Creatinine 196 215 220 224 230 Phosphate 162 175 186 193 208 Vitamin B12 91 100 113 118 131 Clearance: QB: (400ml/min) Urea 290 300 Creatinine 251 259 Phosphate 212 231 Vitamin B12 124 139 The in vitro performance data were obtained with QD = 500ml/min:
QF = 0ml/min; T=37°C (ISO8637)
The ultrafiltration coefficients were maintained using human blood,
Hct = 32%, protein content 6%Effective surface area (m²) 0.8 1.0 1.3 1.6 1.8 2.2 Blood priming volume (ml) 51 63 78 96 113 132 Membrane material Fresenius Polysulfone® Housing material Polycarbonate Potting compound Polyurethane Sterilization method Inline Steam Form of treatment HD Art. No. 5007041 5007051 5007061 5007071 5007081 5007201 -
FX classix (FX60/FX80)
Proven Fresenius quality for standard
hemodialysis
- Proving track record of success of the Helixone® membrane
- Unique FX-class® design
- Purity enhanced — with steam
- High endotoxin retention capacity

Key features
Optimized blood flow conditions
- The lateral blood-inlet port provides a homogenous blood flow in the dialyzer header, preventing stagnation zones. The design minimizes the risk of kinking, contributing to improved safety
- A high capillary density leads to a more even distribution of the fibers. This enables a homogenous distribution of the incoming blood flow into each fiber in the bundle
Homogeneous dialysate flow for better clearances
- The pinnacle structure at both ends of the polypropylene housing together with the potting technology provides an even, radial flow of the dialysate around the individual fibers of the bundle
- The undulation of the hollow fibers prevents dialysate channeling and, thus, enhances the performance of the dialyzer. The higher packing density of the fiber bundle together with the undulation of the hollow fibers enables a uniform dialysate flow within the whole cross-section of fiber bundle
Dialyzer weight
Dialyzer weight is a crucial factor not only in logistics but also in waste management. The housing of FX-class dialyzers is made of polypropylene. In comparison to the widely used polycarbonate it is much lighter with the result that FX-class dialyzers weigh around half as much as most dialyzers. FX 60 classix 107g FX CorDiax 60 107g The benefits of INLINE steam sterilization
No chemical residuals No need for gamma sterilization – high energy ionizing radiation can degrade and alter the material chemistry. Low rinsing volumes Minimal preparation time – since dialyzers are clean on arrival, rinsing times prior to use are substantially reduced. Lower costs Lower rinsing volumes mean reduced preparation times and costs. INLINE steam sterilisation
Purity enhanced — with steam
All FX-class dialyzers are sterilized by the unique INLINE steam sterilization process:
- Both the blood and the dialysate compartment of the dialyzers are rinsed continuously with steam at a temperature of or above 121°C for more a minimum of 15 minutes. Rinsing with hot steam and without chemicals results in extremely low levels of residuals in the dialyzer
- The dialyzer is rinsed with sterile water
- Every dialyzer is tested for fiber integrity using a bubble-point test
- The dialyzers are dried with warm, sterile air
- Finally, after drying the blood inlet and outlet ports are closed
Benefits of the INLINE steam sterilization process
- Highly pure, sterile and pyrogen-free dialyzers without any potentially harmful residuals from sterilization
- Biocompatibility of membranes remains unaffected from sterilization
- Optimized use of resources due to low rinsing volumes: Only 500 mL is required
- Minimized risk of blood leakages and fiber ruptures due to 100% fiber integrity testing
- Dry dialyzers with minimized risk of contamination due to microbial growth
Performance data
FX classix High-Flux dialysers FX 50 classix FX 60 classix FX 80 classix FX 100 classix Clearance (QB = 300 mL/min) Molecular weight (Dalton) Cytochrome c 12,230 55 74 89 100 Inulin 5,200 72 95 113 122 Vitamin B12 1,355 137 162 185 201 Phosphate 132 204 225 244 253 Creatinine 113 224 243 259 264 Urea 60 253 266 279 280 Clearance (QB = 400 mL/min) Cytochrome c 12,230 76 92 105 Inulin 5,200 99 119 129 Vitamin B12 1,355 175 202 222 Phosphate 132 252 279 291 Creatinine 113 277 300 309 Urea 60 312 334 336 Ultrafiltration coeff. (mL/h x mm Hg) 27 38 53 68 Sieving coefficients Albumin 66,500 < 0.001 Myoglobin 17,053 0.1 β2-microglobulin 11,731 0.7 Inulin 5,200 1 In vitro performance: QD = 500 mL/min, QF = 0 mL/min, T = 37°C (ISO8637). Ultrafiltration coefficients: human blood, Hct 32 %, protein content 6 %. Membrane material Helixone Sterilisation method INLINE steam Housing material Polypropylene Potting compound Polyurethane Units per box 24 Effective surface (m²) 1.0 1.4 1.8 2.2 K0A Urea 866 1,068 1,394 1,429 Priming volume (mL) 53 74 95 116 Article number F00002385 F00002386 F00002387 F00002388 -
German-Engineered Gynecological Instruments
German-Engineered
Gynecological Instruments
Handcrafted to exacting standards by the world’s preeminent steel artisans, Euro-Med offers stainless steel specula and biopsy punches, “rose-coated” LEEP instruments, and other specialty instruments for control and precision. Euro-Med instruments are ideal for doctors and practices who use only the best to ensure exceptional results.
EURO-MED Catalog
Stainless Steel and LEEP Coated Instruments for the OB/GYN Office
- Specula
- Specula Dimension Chart
- Lateral Wall Retractors
- Sounds
- Dilators
- Endocervical Curettes
- Endometrial Curettes
- Endocervical Specula
- Tenaculum Forceps
- Cervical Stabilizer
- Hooks
- IUD Hooks
- Biopsy Punches
- Colposcopy Biopsy Instrument Sets
- Vulvar Punch Set
- Scissors
- Hemostatic Forceps
- Uterine Forceps
- Sponge Forceps
- Dressing and Tissue Forceps
- Needle Holders
- LEEP Specula
- LEEP Lateral Wall Retractors
- Potocky Needle and Cartridge Syringe
- LEEP Instruments
- Stainless Steel Care
-
German-Engineered Gynecological Instruments
German-Engineered Gynecological Instruments
Handcrafted to exacting standards by the world’s preeminent steel artisans, Euro-Med offers stainless steel specula and biopsy punches, “rose-coated” LEEP instruments, and other specialty instruments for control and precision. Euro-Med instruments are ideal for doctors and practices who use only the best to ensure exceptional results.
-
GLIDE CATHETER cobra2 (C2)
GLIDECATH® Hydrophilic Coated Catheter
PRODUCT OVERVIEW
GLIDECATH® is a hydrophilically coated, angiographic catheter providing the vessel selectivity and navigation required for a wide variety of diagnostic or interventional procedures.
- Navigate difficult vessel anatomy with smooth tracking facilitated by TERUMO Glide Technology™
- Leverage superior kink resistance to achieve ideal flow rates for diagnostic imaging1,*


SINGLE-BRAIDED
- Reach distal visceral vessels with best-in-class trackability1,*
- Approach tortuous anatomy with best-in-class pushability1,*
*When compared to Merit Impress and AngioDynamics Mariner during mechanical bench testing

DOUBLE-BRAIDED
- Access and cross challenging lesions with excellent pushability and torquability1
- Achieve superior device delivery through a nylon-rich inner layer1,*
- Leverage superior kink resistance to maintain lumen integrity throughout the procedure1,*
TIP SHAPE PORTFOLIO
GLIDECATH® is offered in 17 unique tip shapes that are designed for various anatomy.

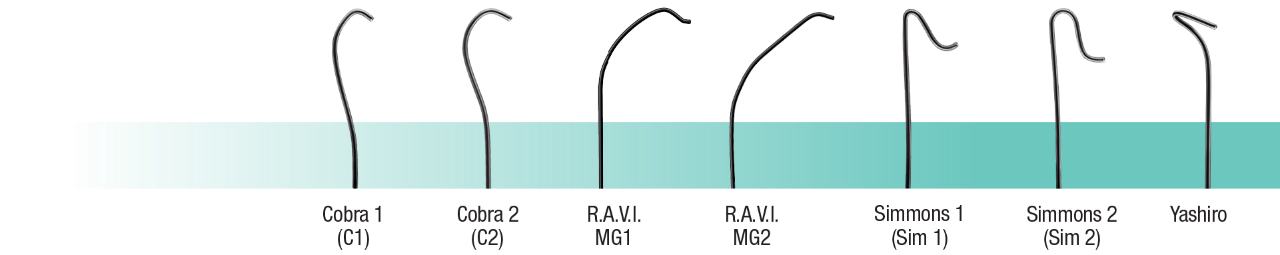

PRODUCT CODES
GLIDECATH® 4 Fr Product Codes
TIP SHAPES CATHETER LENGTH 4 Fr 65 cm 70 cm 100 cm 120 cm 125 cm 135 cm 150 cm Straight CG412 – CG413 CG414 – – CG431 Angle CG415 – CG416 CG417 – – CG432 C1 CG408 – – – – – – C2 CG409 – CG410 – – – CG434 J-Tip CG411 – – – – – – Yashiro* – CG422 – – – – – Sim 1 – – CG401 – – – – Sim 2 – – CG402 – – – – Sim 3 – – CG403 – – – – H1 – – CG404 – – – – JB1 – – CG405 – – – – JB2 – – CG406 – – – – JR 4.0 – – – – – – CG435 RAVI MG1 – – – – CG440 – – RAVI MG2 – – – – – CG441 CG442 MP – – CG418 – – – – Mani – – CG407 – – – – Pigtail – – – – – – CG433 PV Multicurve – – – – – – CG430 GLIDECATH® 5 Fr Product Codes
TIP SHAPES CATHETER LENGTH 5 Fr 65 cm 70 cm 100 cm 120 cm 125 cm 135 cm 150 cm Straight CG505 – CG506 – – – – Angle Taper CG507 – CG508 – – – – C1 CG501 – – – – – C2 CG502 – CG503 – – – – J-Tip CG504 – – – – – – Yashiro* – CG519 – – – – – Sim 1 – – CG510 – – – – Sim 2 CG521 – CG511 – – – – Sim 3 – – CG512 – – – – H1 – – CG513 – – – – JB1 – – CG514 – – – – JB2 – – CG515 – – – – MP – – CG509 – – – – Mani – – CG516 – – – – GLIDECATH® 5 Fr XP Product Codes
TIP SHAPES CATHETER LENGTH 5 Fr XP 65 cm 70 cm 100 cm 120 cm 125 cm 135 cm 150 cm Straight RF*ZM7510GM – RF*ZM75110M – – – RF*ZM75115JM Angle RF*ZV9510GM – RF*ZV95110M – – – – C2 RF*ZB5510GM – – – – – Sim 1 – – RF*WA15110M – – – – Sim 2 – – RF*WA25110M – – – – H1 – – RF*WB15110M – – – – RAVI MG1 – – – – RF*ZRV511CM – – RAVI MG2 – – – – – RF*ZUF511DJM RF*ZUF5115JM PRODUCT SPECIFICATIONS
4 Fr 5 Fr 5 Fr XP Effective Lengths 65 cm, 70 cm, 100 cm, 120 cm, 135 cm & 150 cm 65cm, 70cm & 100cm 65 cm, 100 cm, 125 cm, 135 cm & 150 cm Construction Double-Braided Stainless Steel Single-Braided Stainless Steel Double-Braided Stainless Steel Inner Diameter – Distal Tip 0.0405” / 1.03 mm 0.0433” / 1.10 mm 0.0472” / 1.20 mm Inner Diameter – Proximal Shaft 0.0413” / 1.05 mm 0.0441” / 1.12 mm 0.0480” / 1.22 mm Guidewire Compatibility 0.038″ 0.038″ 0.038″ Maximum Injection Pressure 750 psi 1000 psi 1000 psi Shape Straight
Angle
Cobra 1 (C1)
Cobra 2 (C2)
J-Tip
Yashiro
Simmons 1 (SIM1)
Simmons 2 (SIM2)
Simmons 3 (SIM3)
Headhunter
Berenstein 1 (BERN 1)
Berenstein 2 (BERN 2)
Multi-Purpose
Mani
PV Multicurve
R.A.V.I. MG1
R.A.V.I. MG2Straight
Angle
Cobra 1 (C1)
Cobra 2 (C2)
J-Tip
Yashiro
Simmons 1 (SIM1)
Simmons 2 (SIM2)
Simmons 3 (SIM3)
Headhunter
Berenstein 1 (BERN1)
Berenstein 2 (BERN2)
Multi-Purpose
ManiStraight
Angle
Cobra 2 (C2)
Simmons 1 (SIM1)
Simmons 2 (SIM2)
Headhunter
R.A.V.I. MG1
R.A.V.I. MG2Hydrophilic Coating Distal 15 cm & Distal 40 cm Distal 25 cm & Distal 40 cm Distal 15 cm & Distal 40 cm -
Glide sheath
Product overview
Expanding trans-radial access to include more patients
- Glide sheath Slender is designed with a proprietary thin-wall technology where the outside diameter is reduced by one French size while the inner-diameter equivalent is maintained.
- Glide sheath Slender is coated with Terumo’s proprietary hydrophilic coating, allowing for smooth insertion and removal.
Features and Benefits

The proprietary thin-wall technology and Terumo M Coat hydrophilic coating
- Per the American Heart Association, dedicated radial sheaths with hydrophilic coating combined with smaller diameter sheaths and a tapered tip design may reduce risk of arterial spasm and radial occlusion1
- Featuring Terumo’s proprietary hydrophilic coating, the Glide sheath Slender Sheath is the only thin-wall radial sheath on the market to demonstrate superior and consistent performance in lubricity and durability over time
The Glide sheath Slender Sheath is the only thin-wall sheath that demonstrated best in class performance in all of the American Heart Association’s identified critical features that lead to reduced spasm.
THE POWER OF TERUMO GLIDE TECHNOLOGY™: LUBRICITY OVER TIMES
Hydrophilic coating is critical to procedural success, Physicians need to trust that their sheath will be just as lubricous at the end of case as it is at the beginning.
The graph below demonstrates the results of recent benchtop testing. The lower the value, the more lubricious the coating. The straighter the line, the more consistent, and reliable, the performance.
Glide sheath Slender Sheath outperformed the other competitive sheaths in both key features: lubricity and lubricity over time.

Unmatched Clinical Evidence
- A meta-analysis of 10 clinical papers, involving 3,821 patients, demonstrated that use of Glide sheath Slender Sheath is associated with high rates of procedural success and low rates in performance related issues.
- In studies measuring patient comfort, patients who underwent percutaneous trans-radial procedures with the Glide sheath Slender Sheath reported none to low amounts of pain.
-
Glidepath Long term Hemodialysis Catheter with Preloaded Stylet Standard Kit (14.5F)(Straight) 15 cm
AVAILABEL SIZE-
6393150
Glidepath Long term Hemodialysis Catheter with Preloaded Stylet Standard Kit (14.5F)(Straight) 15 cm
6393270
Glidepath Long term Hemodialysis Catheter with Preloaded Stylet Standard Kit (14.5F)(Straight) 27 cm
6396190
Glidepath Long term Hemodialysis Catheter with Preloaded Stylet Standard Kit (14.5F)(Alpha) 19 cm
6396240
Glidepath Long term Hemodialysis Catheter with Preloaded Stylet Standard Kit (14.5F)(Alpha) 24 cm
6393190
Glidepath Long term Hemodialysis Catheter with Preloaded Stylet Standard Kit (14.5F)(Straight) 19 cm
6393230
Glidepath Long term Hemodialysis Catheter with Preloaded Stylet Standard Kit (14.5F)(Straight) 23 cm
6393310
GlidePath Long-Term Hemodialysis Catheter with Preloaded Stylet Standard Kit (14.5F)(Straight)(31cm)
6393350
GlidePath Long-Term Hemodialysis Catheter with Preloaded Stylet Standard Kit (14.5F)(Straight)(35cm)
6393420
GlidePath Long-Term Hemodialysis Catheter with Preloaded Stylet Standard Kit (14.5F)(Straight)(42cm)
6396280
GlidePath Long-Term Hemodialysis Catheter Standard Kit (14.5F)(Alphacurve Configuration) (28cm)
-
GRAFT INTERGARD WOVEN STRAIGHT
Intergard Woven
Woven Vascular Grafts
- Collagen-coated polyester graft
- External velour surface and non-velour inner surface
- Magnetic Resonance Safe
- Water permeability : < 5ml/cm²/min[1]
Straight
Diameter Length Reference Diameter Length Reference Diameter Length Reference 12 mm 15 cm IGW0012-15 6 mm 30 cm IGW0006-30 22 mm 60 cm IGW0022-60 14 mm 15 cm IGW0014-15 8 mm 30 cm IGW0008-30 24 mm 60 cm IGW0024-60 16 mm 15 cm IGW0016-15 10 mm 30 cm IGW0010-30 26 mm 60 cm IGW0026-60 18 mm 15 cm IGW0018-15 12 mm 30 cm IGW0012-30 28 mm 60 cm IGW0028-60 20 mm 15 cm IGW0020-15 14 mm 30 cm IGW0014-30 30 mm 60 cm IGW0030-60 22 mm 15 cm IGW0022-15 16 mm 30 cm IGW0016-30 32 mm 60 cm IGW0032-60 24 mm 15 cm IGW0024-15 18 mm 30 cm IGW0018-30 26 mm 15 cm IGW0026-15 20 mm 30 cm IGW0020-30 28 mm 15 cm IGW0028-15 22 mm 30 cm IGW0022-30 30 mm 15 cm IGW0030-15 24 mm 30 cm IGW0024-30 32 mm 15 cm IGW0032-15 26 mm 30 cm IGW0026-30 34 mm 15 cm IGW0034-15 28 mm 30 cm IGW0028-30 36 mm 15 cm IGW0036-15 30 mm 30 cm IGW0030-30 32 mm 30 cm IGW0032-30 34 mm 30 cm IGW0034-30 36 mm 30 cm IGW0036-30 38 mm 30 cm IGW0038-30 
Bifurcated
Diameter Length Reference 12 mm x 6 mm 50 cm IGW1206 14 mm x 7 mm 50 cm IGW1407 16 mm x 8 mm 50 cm IGW1608 18 mm x 9 mm 50 cm IGW1809 20 mm x 10 mm 50 cm IGW2010 22 mm x 11 mm 50 cm IGW2211 24 mm x 12 mm 50 cm IGW2412 -
GRAFTMASTER
GRAFTMASTER
RX Coronary Stent Graft System
HDE INDICATION
The GRAFTMASTER™ RX Coronary Stent Graft System is indicated for use in the treatment of free perforations, defined as free contrast extravasation into the pericardium, in native coronary vessels or saphenous vein bypass grafts ≥ 2.75 mm in diameter. The effectiveness of this device for this use has not been demonstrated. Long-term outcome for this permanent implant is unknown at present.
HUMANITARIAN USE DEVICE
The effectiveness of this device for this use has not been demonstrated. IRB approval required for use.
GRAFTMASTER™ DIAMETER 2.8 – 4.0 MM 4.5 – 4.8 MM Stent Material Stainless Steel 316L Stainless Steel 316L Graft Material Expandable Polytetrafluoroethylene (ePTFE)
sandwiched between two identical stentsExpandable Polytetrafluoroethylene (ePTFE)
sandwiched between two identical stentsDouble Wall Thickness (mm) 0.52 0.52 Balloon Type Semi Compliant Semi Compliant Shaft Size (F) 2.4 – 2.8 2.4 – 2.8 Nominal Deployment Pressure (ATM) 15 15 Rated Burst Pressure (ATM) 16 16 Maximum Guide Wire (in) 0.014 0.014 Total Length (cm) 143 143 Maximum Stent Graft Expansion (mm)* 5.5 5.5 Minimum Guide Catheter (in) / (F) 0.068 / ≥ 6 0.074 / ≥ 7 Crimped Stent Profile (in) 0.064 0.068 Tip Entry Profile (in) 0.024 0.024 Maximum Crossing Profile (in) 0.064 0.068 Product specification data on file at Abbott.
*The length of the ePTFE foil is shorter than the reported strut length.
After expansion of the stent, the ePTFE foil may be up to 1.6 mm from each end of the stent graft. The covered length of the stented area may be up to 3.2 mm shorter than the stent length. This information must be considered when choosing an appropriately sized stent graft.
COMPLIANCE CHART
Inner Diameter Compliance Chart
ATM 2.80 MM 3.50 MM 4.00 MM 4.50 MM 4.80 MM 11 1.37 1.83 2.30 2.86 2.98 12 1.67 2.34 2.74 3.20 3.57 13 1.91 2.47 2.90 3.30 3.89 14 2.08 2.63 3.08 3.52 3.98 15 (NOM) 2.18 2.81 3.31 3.79 4.16 16 (RBP) 2.32 3.01 3.54 3.98 4.34 17 2.47 3.19 3.72 4.15 4.52 18 2.62 3.32 3.86 4.31 4.64 19 2.73 3.43 3.96 4.42 4.77 Outer Diameter Compliance Chart
ATM 2.80 MM 3.50 MM 4.00 MM 4.50 MM 4.80 MM 11 1.89 2.35 2.82 3.38 3.50 12 2.19 2.86 3.26 3.72 4.09 13 2.43 2.99 3.42 3.82 4.41 14 2.60 3.15 3.60 4.04 4.50 15 (NOM) 2.70 3.33 3.83 4.31 4.68 16 (RBP) 2.84 3.53 4.06 4.50 4.86 17 2.99 3.71 4.24 4.67 5.00 18 3.14 3.84 4.38 4.83 5.16 19 3.25 3.95 4.48 4.94 5.29 Balloon pressures should be monitored during inflation. Do not exceed rated burst pressure as indicated on product label. Use of pressures higher than specified on the product label may possibly result in a ruptured balloon and potential intimal damage and dissection.
ORDERING INFORMATION
HUMANITARIAN USE DEVICE
The effectiveness of this device for this use has not been demonstrated.
IRB approval required for use.GRAFTMASTER™ RX Stent Graft
Diameter (mm)Stent Graft Length (mm) 6F/7F
(Guide)16 19 26 2.8 1012580-16 1012580-19 1012580-26 6 3.5 1012581-16 1012581-19 1012581-26 6 4.0 1012582-16 1012582-19 1012582-26 6 4.5 1012583-16 1012583-19 1012583-26 7 4.8 1012584-16 1012584-19 1012584-26 7 MAT-2201048 v1.0
IMPORTANT SAFETY INFORMATION
 GRAFTMASTER™ RX
GRAFTMASTER™ RX
Coronary Stent Graft SystemHumanitarian Device
Humanitarian Device. Authorized by Federal Law for the use in the treatment of free perforations, defined as free contrast extravasation into the pericardium, in native coronary vessels or saphenous vein bypass grafts ≥ 2.75 mm in diameter. The effectiveness of this device for this use has not been demonstrated.
Indications
The GRAFTMASTER™ RX is indicated for use in the treatment of free perforations, defined as free contrast extravasation into the pericardium, in native coronary vessels or saphenous vein bypass grafts ≥ 2.75 mm in diameter. The effectiveness of this device for this use has not been demonstrated. Long-term outcome for this permanent implant is unknown at present.
Contraindications
GRAFTMASTER™ RX is contraindicated for use in:
- Patients in whom antiplatelet and/or anticoagulation therapy is contraindicated.
- Patients who are judged to have a treatment area that prevents complete inflation of an angioplasty balloon or proper placement of the stent graft.
Warnings
- Ensure that the sterile barrier has not been opened or damaged prior to use.
- Judicious selection of patients is necessary, since the use of this device carries the associated risk of subacute thrombosis, vascular complications, and / or bleeding events.
- Persons allergic to 316L stainless steel (including the major elements iron, chromium, nickel, molybdenum) or PTFE may suffer an allergic reaction to this implant.
- When multiple stents are required, stent materials should be of similar composition. Placing multiple stents of different metals in contact with each other may increase the potential for corrosion. The risk of in vivo corrosion does not appear to increase based on in vitro corrosion tests using an L-605 CoCr alloy stent (MULTI-LINK VISION® Coronary Stent) in combination with a 316L stainless steel alloy stent (MULTI-LINK TETRA Coronary Stent).
Precautions
General Precautions
(See also Individualization of Treatment in IFU)
- Implantation of the stent graft should be performed only by physicians who have received appropriate training.
- Subsequent restenosis may require repeat dilatation of the arterial segment containing the stent graft. The long-term outcome following repeat dilatation of endothelialized stent grafts is unknown at present.
- Care should be taken to control the guiding catheter tip during stent graft delivery, deployment, and balloon withdrawal. Before withdrawing the stent graft delivery system, visually confirm complete balloon deflation by fluoroscopy to avoid guiding catheter movement into the vessel and subsequent arterial damage.
- Carefully read all instructions prior to use. Observe all warnings and precautions noted throughout these instructions. Failure to do so may result in complications.
- Note the product “Use by” date specified on the package.
Stent Graft Handling – Precautions
- This device is intended for single-use only; do not reuse. Do not resterilize, as this can compromise the device performance and increase the risk of cross contamination due to inappropriate reprocessing.
- Do not remove the Stent Graft from its Delivery System. Removing the Stent Graft from the Delivery System may damage the Stent Graft and/or lead to Stent Graft embolization.
- The delivery system should not be used in conjunction with other stents.
- Special care must be taken not to handle or in any way disrupt the Stent Graft position on the Delivery System. This is most important during placement over the guide wire and advancement through the hemostasis valve adapter and guiding catheter hub.
- Excessive manipulation (e.g., rolling the mounted Stent Graft) may cause dislodgment of the Stent Graft from the delivery balloon.
- Do not manipulate, touch, or handle the stent graft with your fingers, as this may cause contamination or dislodgement of the stent graft from the delivery balloon.
- Use only appropriate balloon inflation media. Do not use air or any gas medium to inflate the balloon as it may cause uneven expansion and difficulty in deployment of the Stent Graft.
Stent Graft Placement – Precautions
Stent Graft Preparation – Precautions
- Do not prepare or pre-inflate balloon prior to stent graft deployment other than as directed. Use the balloon purging technique described in Section 10.2.3 of the GRAFTMASTER™ RX IFU Delivery System Preparation.
- While introducing the delivery system into the vessel, do not induce negative pressure on the delivery system. This may cause dislodgement of the stent graft from the balloon.
- Use guiding catheters which have lumen sizes that are suitable to accommodate the stent graft delivery system (See section 10.1 of the GRAFTMASTER™ RX IFU – Materials Required or product label).
Stent Graft Implantation – Precautions
- Pre-dilatations of the vessel must take into account proximal atherosclerotic plaque beyond the treatment area, which may prevent advancement of the device to the treatment area. Failure to do so may increase the difficulty of stent graft placement and cause procedural complications.
- Implanting a stent graft may lead to dissection of the vessel distal and / or proximal to the stent graft, and may cause closure of the vessel, requiring additional intervention (e.g., coronary artery bypass surgery, further dilatation, placement of additional stents, etc.).
- If more than one Stent Graft is required, the distal Stent Graft should be placed initially, followed by placement of the proximal Stent Graft. Stent Graft placement in this order obviates the need to cross the proximal Stent Graft when placing the distal Stent Graft, and reduces the chances for dislodging the proximal Stent Graft.
- Do not expand the Stent Graft if it is not properly positioned in the vessel (See Stent Graft/System Removal – Precautions)
- Placement of the Stent Graft will compromise side-branch patency.
- Do not exceed the rated burst pressure (RBP) as indicated on the product label. Monitor balloon pressures during inflation. Use of pressures higher than specified on the product label may result in a ruptured balloon with possible intimal damage and dissection.
- An unexpanded stent graft may be retracted into the guiding catheter one time only. An unexpanded stent graft should not be reintroduced into the artery once it has been pulled back into the guiding catheter. Subsequent movement in and out through the distal end of the guiding catheter should not be performed, as the stent graft may be damaged when retracting the undeployed stent graft back into the guiding catheter.
- Stent graft retrieval methods (use of additional wires, snares, and / or forceps) may result in additional trauma to the vasculature and / or the vascular access site. Complications may include bleeding, hematoma, or pseudoaneurysm.
Stent Graft/System Removal – Precautions
Removal of the delivery System Prior to Stent Graft Deployment:
- If removal of the stent graft system is required prior to deployment, ensure that the guiding catheter is coaxially positioned relative to the stent graft delivery system, and cautiously withdraw the stent graft delivery system into the guiding catheter.
- Should unusual resistance be felt at any time, either during lesion access or during removal of the Delivery System post- Stent Graft implantation, the Delivery System and guiding catheter should be removed as a single unit. This must be done under direct visualization of fluoroscopy.
Withdrawal of the Stent Graft Delivery System from the Deployed Stent Graft
- Deflate the balloon by pulling negative on the inflation device. Confirm balloon deflation under fluoroscopy and wait 10 – 15 seconds longer.
- Position the inflation device to “negative” or “neutral” pressure.
- Stabilize guide catheter position just outside coronary ostium and anchor in place. Maintain guide wire placement across the stent graft segment.
- Gently remove the stent graft delivery system with slow and steady pressure.
- Tighten the rotating hemostatic valve.
Note: If, during withdrawal of the catheter, resistance is encountered, use the following steps to improve balloon rewrap:
- Re-inflate the balloon up to nominal pressure.
- Repeat steps 1 through 5 above.
- Failure to follow these steps and / or applying excessive force to the delivery system can potentially result in loss or damage to the stent graft and / or delivery system components.
- If it is necessary to retain guide wire position for subsequent artery / treatment area access, leave the guide wire in place and remove all other system components.
- Retrieval methods (i.e., additional wires, snares, and / or forceps) may result in additional trauma to the coronary vasculature and / or the vascular access site. Complications may include, but are not limited to, bleeding, hematoma, or pseudoaneurysm.
Post-Stent Graft Placement – Precautions
- Care must be exercised when crossing a newly deployed stent graft with an intravascular ultrasound (IVUS) catheter, a coronary guide wire, a balloon catheter, or delivery system to avoid disrupting the stent graft geometry, apposition, and / or geometry.
- Antiplatelet therapy should be administered post-procedure (See Individualization of Treatment in IFU). Patients who require early discontinuation of antiplatelet therapy (e.g., secondary to active bleeding) should be monitored carefully for cardiac events. At the discretion of the patient’s treating physician, the antiplatelet therapy should be restarted as soon as possible.
- If the patient requires imaging, see MRI Statement.
MRI Statement
- Nonclinical testing has demonstrated that the GRAFTMASTER™ RX Coronary Stent Graft, in single and in overlapped configurations up to 44 mm in length, is MR Conditional. It can be scanned safely under the following conditions:
- Static magnetic field of 1.5 or 3 Tesla
- Spatial gradient field of 2500 Gauss/cm or less
- Maximum whole-body-averaged specific absorption rate (SAR) of 2.0 W/kg (normal operating mode) for up to 15 minutes of scanning for each duration of a sequence
- The GRAFTMASTER™ RX stent graft should not migrate in this MRI environment. Nonclinical testing at field strengths greater than 3 Tesla has not been performed to evaluate stent graft migration or heating. MRI at 1.5 or 3 Tesla may be performed immediately following the implantation of the GRAFTMASTER™ RX stent graft.
- Stent graft heating was derived by using the measured nonclinical, in vitro temperature rises in a GE Excite 3 Tesla scanner and in a GE 1.5 Tesla coil in combination with the local specific absorption rates (SARs) in a digitized human heart model. The maximum whole body averaged SAR was determined by validated calculation. At overlapped lengths of up to 44 mm, the GRAFTMASTER™ RX stent graft produced a nonclinical maximum local temperature rise of 1.8ºC at a maximum whole body averaged SAR of 2.0 W/kg (normal operating mode) for 15 minutes. These calculations do not take into consideration the cooling effects of blood flow.
- The effects of MRI on overlapped stent grafts greater than 44 mm in length or stent grafts with fractured struts are unknown.
- As demonstrated in nonclinical testing, the image artifact extends approximately 15 mm from the device, both inside and outside the device lumen, when scanned using the sequence: gradient echo in a 3T GE Sigma HDxt software release 15.0_M4_0910.z MR system with a Body Transmit coil. Therefore, it may be necessary to optimize the MR imaging parameters for the presence of the GRAFTMASTER™ RX stent graft.
- It is suggested that patients register the conditions under which the implant can be safely scanned with the MedicAlert Foundation (medicalert.org) or an equivalent organization.
Potential Adverse Events
Adverse events (in alphabetical order) that may be associated with the use of GRAFTMASTER™ RX Coronary Stent Graft in native coronary arteries may include:
- Acute myocardial infarction
- Arrhythmias (including ventricular fibrillation and ventricular tachycardia)
- Coronary Artery Bypass Surgery
- Death
- Dissection
- Drug reactions to antiplatelet agents/contrast medium
- Emboli, distal (air, tissue or thrombotic emboli)
- Emergent Coronary Artery Bypass Surgery
- Hemorrhage, requiring transfusion
- Hypotension / Hypertension
- Infection and pain at insertion site
- Ischemia, myocardial
- Perforation
- Pseudoaneurysm, femoral
- Restenosis of stented segment
- Spasm
- Stent graft embolization
- Stent Graft thrombosis / occlusion
- Stroke/Cerebrovascular Accidents
- Total occlusion of coronary artery