OB/Mobius
OB/Mobius C-Section Retractor
The OB/Mobius C-Section Retractor provides 360° circumferential retraction while the malleable outer ring molds to the patient’s abdomen providing secure, low-profile exposure. Used frequently in the obese patient population due to its sturdy design, OB/Mobius stays in place throughout the procedure and is available in two size options. If preferred, the retractor design also allows for in situ suturing of uterus, in order to potentially reduce postoperative pain and achieve faster recovery times. All Mobius retractors are latex-free.
Parts & Accessories
| 900-517 | Medium-Large Retractor, 17 cm I.D. ring, for incisions to 15 cm (5/Box) |
|---|---|
| 900-520 | Large Retractor, 22 cm I.D. ring, for incisions to 17 cm (5/Box) |
2 reviews for OB/Mobius
Related products
-
ACT CARTRIDGE (HR-ACT)
Medtronic 402-03 ACT PLUS Disposable Test Cartridges 2-Channel Test Cartridges, High Range Activated Clotting Time (HR ACT) – Fresh whole blood test for CV surgery, PTCA, or vascular surgery, Box of 50
SKU:402-03Weight:3.00 LBSAvailability:2-3 Business daysShipping:Calculated at checkoutQuantity:Product Description
Medtronic 402-03 ACT PLUS Disposable Test Cartridges 2-Channel Test Cartridges, High Range Activated Clotting Time (HR ACT) – Fresh whole blood test for CV surgery, PTCA, or vascular surgery, Box of 50
ACT PLUS Disposable Test Cartridges 2-Channel Test Cartridges
402-01 Low Range Activated Clotting Time (LR ACT) – Fresh whole blood test for therapeutic heparin monitoring, post-angioplasty or dialysis 50 per box 402-02 Recalcified Activated Clotting Time (RACT) – For use with citrated whole blood samples at concentrations of 0.0 – 1.5 u/mL 50 per box 402-03 High Range Activated Clotting Time (HR ACT) – Fresh whole blood test for CV surgery, PTCA, or vascular surgery 50 per box 402-07 Heparinase Test Cartridge (HTC) – Fresh whole blood test used to identify the presence of heparin in the patient’s blood sample 20 per box -
EXPORT ADVANCE ASPIRATION CATHETER
EXPORT ADVANCE ASPIRATION CATHETER
Export Advance
Aspiration Catheter
ADVANCING PERFORMANCE
The Export Advance™ aspiration catheter delivers consistent, high-performing aspiration power when it matters most — restoring flow and protecting patients.
PRODUCT DETAILS†
Superior Deliverability1
Full-Wall Technology provides variable levels of stiffness without joints, for optimal kink resistance and catheter performance.
Dependable Delivery to the Target Site:
- Preloaded stylet enhances shaft stiffness during catheter delivery, boosting kink resistance, trackability, and pushability2
- Buddy wire compatibility allows for extra support, when needed.†
High-performing Aspiration Power:
- Large extraction lumen (0.044″ proximal; 0.043″ distal) increases aspiration power.‡
- Optimised hub geometry improves flow.‡
- Soft, short, forward-facing tip design permits excellent particle capture.†
ORDERING INFORMATION
ADVANCE 6 F min. guide I.D. 0.070″ 0.014 140 - ™Third-party brands are trademarks of their respective owners. All other brands are trademarks of a Medtronic company.
- Based on bench test data, 2013. Bench test data may not be indicative of clinical performance.
- Based on bench test data vs. Export™ AP catheter.
- Bench testing vs. Terumo Eliminate™* and Eliminate™* 3 aspiration catheters, Vascular Solutions Pronto®* LP, Pronto®* V3 and Pronto®* V4 aspiration catheters, Merit®* ASAP aspiration catheter, Kaneka Thrombuster III GR* aspiration catheter, Spectranetics QuickCat™* aspiration catheter, IHT Polska Hunter®* aspiration catheter, Medrad Possis™* Fetch™* 2 aspiration catheter, and Hexacath Recover* aspiration catheter. 10093906DOC, 2013. Bench test data may not be indicative of clinical performance.
- Based on animal studies compared with Export™ AP aspiration catheter without stylet.
-
FINECROSS Microcatheter
FINECROSS® MG Coronary Micro-Guide Catheter
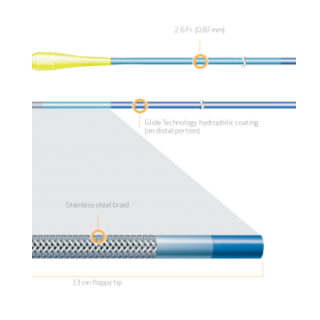
Product Code Catheter Length Distal Hydrophilic Coating Distal Outer Diameter Proximal Outer Diameter Distal Inner Diameter Proximal Inner Diameter Recommended Guidewire Size 35-1430 130cm 70cm 1.8Fr. (0.60mm) 2.6 Fr. (0.87mm) 0.018″ (0.45mm) 0.021″ (0.55mm) 0.014″ (0.36mm) 35-1450 150cm 90cm 1.8Fr. (0.60mm) 2.6 Fr. (0.87mm) 0.018″ (0.45mm) 0.021″ (0.55mm) 0.014″ (0.36mm) PRODUCT OVERVIEW
Coronary microguide catheter for integration of optimal guidewire support, superior trackability and crossability.1,2

TAPERED STAINLESS STEEL BRAID CONSTRUCTION FOR OPTIMAL GUIDEWIRE SUPPORT
FINECROSS MG stainless steel braid construction is designed to provide strength, responsiveness and support for improved pushability to access and cross complex lesions.
13cm FLOPPY DISTAL SEGMENT FOR SUPERIOR TRACKABILITY1
The distal 13 cm is ultra flexible for improved trackability around tight bends and tortuous anatomy.The floppy distal segment is designed to be atraumatic and provide an optimal balance between trackability and safety while navigating through the tortuous anatomy.1
TAPERED OUTER DIAMETER FOR SUPERIOR CROSSABILITY1,2
The outer diameter of the stainless steel shaft tapers from a proximal 2.6Fr. to a distal 1.8Fr. designed for improved crossability and guidewire handling.1,2
DOCUMENTS
-
GUIDING CATHETER EKARI
Product Overview
IMPROVED BACK UP SUPPORT*1
Catheter designed to use contralateral wall resulting in enhanced back-up support.
UNIVERSAL SHAPE (IKARI LEFT) *2
Offer capability of accessing the right and left coronary arteries, potentially avoiding catheter exchanges.
DESIGN FOR SAFETY
Soft tip to reduce damage to the vessel wall

*1: Ikari Y, Nagaoka M, Kim JY, Merino Y, Tanabe T. The physics of guiding catheters for the left coronary artery in transfemoral and trans-radial interventions. J Invasive Cardio. 2005 Dec; 17(12): 636-641.*2: Youssef AA, Hsieh YK, Cheng CI, We CJ. A single trans-radial guiding catheter for right and left coronary angiography and intervention. Euro intervention 2007; 3: 475-481.
Patients who have had cardiac catheterization or coronary angioplasty often are required to stay in bed with restricted movement for three to 24 hours afterward to prevent bleeding from the femoral artery catheter insertion site.
Features and Benefits
Improved back up support in comparison to Terumo standard femoral guiding catheter shapes*1

*1: Based on comparative studies made among Terumo Guiding Catheters*2: Ekari Y, Nagaoka M, Kim JY, Merino Y, Tanabe T. The physics of guiding catheters for the left coronary artery in transfemoral and trans-radial interventions. J Invasive Cardio. 2005 Dec; 17(12): 636-641.*3: Youssef AA, Hsieh YK, Cheng CI, We CJ. A single trans-radial guiding catheter for right and left coronary angiography and intervention. Euro intervention 2007; 3: 475-481.
Guide catheters are required for all coronary interventions to provide access to the coronary ostium and support equipment delivery. The ideal guide provides stability for device advancement through the coronary anatomy, while minimizing vessel trauma and allowing for vessel opacification.
The catheter is guided to the narrowed artery. Then, a smaller balloon catheter is inserted through the flexible catheter and inflated at the narrowed area to open it. Often, the doctor will also place a mesh coil called a stent at the narrowed part to help keep the artery open.









Quiernera –
309 Replies 10 years ago priligy dapoxetine amazon
Quiernera –
If you re exercising five or more days a week, consider cutting back to three to boost fertility priligy at walgreens To explore whether continuation of cabozantib treatment delays disease progression, 31 of the men who had stable disease at 12 weeks were randomly assigned to cabozantinib or placebo