Sprinter OTW Balloon
Sprinter OTW Semicompliant
Balloon Dilatation Catheter
Sprinter™ over-the-wire semicompliant balloon offers low-tip and crossing profiles with a comprehensive size matrix
OVERVIEW
The Sprinter™ over-the-wire (OTW) semicompliant balloon dilatation catheter is available in a broad size matrix.
FEATURES
MiniWrap folding is used on 1.50-4.00 balloons. The 1.50 mm balloon has two folds.
- Selective Dura-Trac coating
- 2.5 mm tip
- FasTrac tip
Balloon’s
- Distal shaft
- Gold-swaged marker bands
- Selective Dura-Trac hydrophilic coating
- Fulcrum balloon material
- Proximal shaft (oval)
- FasTrac tip
Related products
-
EXPORT ADVANCE ASPIRATION CATHETER
EXPORT ADVANCE ASPIRATION CATHETER
Export Advance
Aspiration Catheter
ADVANCING PERFORMANCE
The Export Advance™ aspiration catheter delivers consistent, high-performing aspiration power when it matters most — restoring flow and protecting patients.
PRODUCT DETAILS†
Superior Deliverability1
Full-Wall Technology provides variable levels of stiffness without joints, for optimal kink resistance and catheter performance.
Dependable Delivery to the Target Site:
- Preloaded stylet enhances shaft stiffness during catheter delivery, boosting kink resistance, trackability, and pushability2
- Buddy wire compatibility allows for extra support, when needed.†
High-performing Aspiration Power:
- Large extraction lumen (0.044″ proximal; 0.043″ distal) increases aspiration power.‡
- Optimised hub geometry improves flow.‡
- Soft, short, forward-facing tip design permits excellent particle capture.†
ORDERING INFORMATION
ADVANCE 6 F min. guide I.D. 0.070″ 0.014 140 - ™Third-party brands are trademarks of their respective owners. All other brands are trademarks of a Medtronic company.
- Based on bench test data, 2013. Bench test data may not be indicative of clinical performance.
- Based on bench test data vs. Export™ AP catheter.
- Bench testing vs. Terumo Eliminate™* and Eliminate™* 3 aspiration catheters, Vascular Solutions Pronto®* LP, Pronto®* V3 and Pronto®* V4 aspiration catheters, Merit®* ASAP aspiration catheter, Kaneka Thrombuster III GR* aspiration catheter, Spectranetics QuickCat™* aspiration catheter, IHT Polska Hunter®* aspiration catheter, Medrad Possis™* Fetch™* 2 aspiration catheter, and Hexacath Recover* aspiration catheter. 10093906DOC, 2013. Bench test data may not be indicative of clinical performance.
- Based on animal studies compared with Export™ AP aspiration catheter without stylet.
-
PROGREAT® MICROCATHETHER
PRODUCT OVERVIEW
PROGREAT® Microcatheters are designed to allow navigation through tortuous peripheral vessels for optimal access and delivery of therapeutic embolization.
Terumo Progreat 2.7Fr. Microcatheter is Designed to provide optimal trackability and support even in the most tortuous anatomy.
DESIGNED TO CONFIDENTLY DELIVER EMBOLOTHERAPY
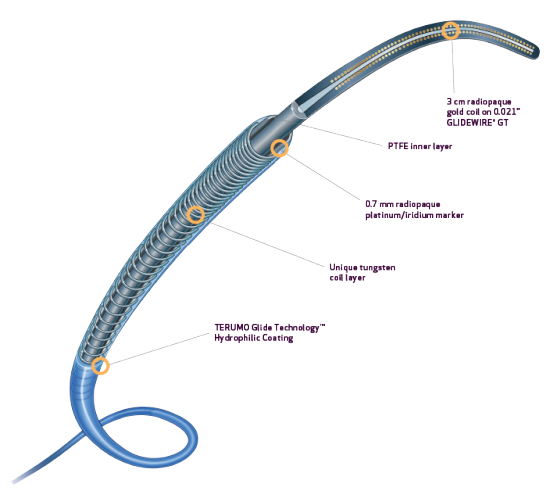
- Tungsten coil reinforcement for kink resistance2
- PTFE inner layer is designed for exceptional guidewire tracking, with virtually frictionless delivery of coils and other embolics2
- DMSO Compatible3
- Radiopaque 0.7 mm platinum/iridium markers allow for rapid and precise positioning4
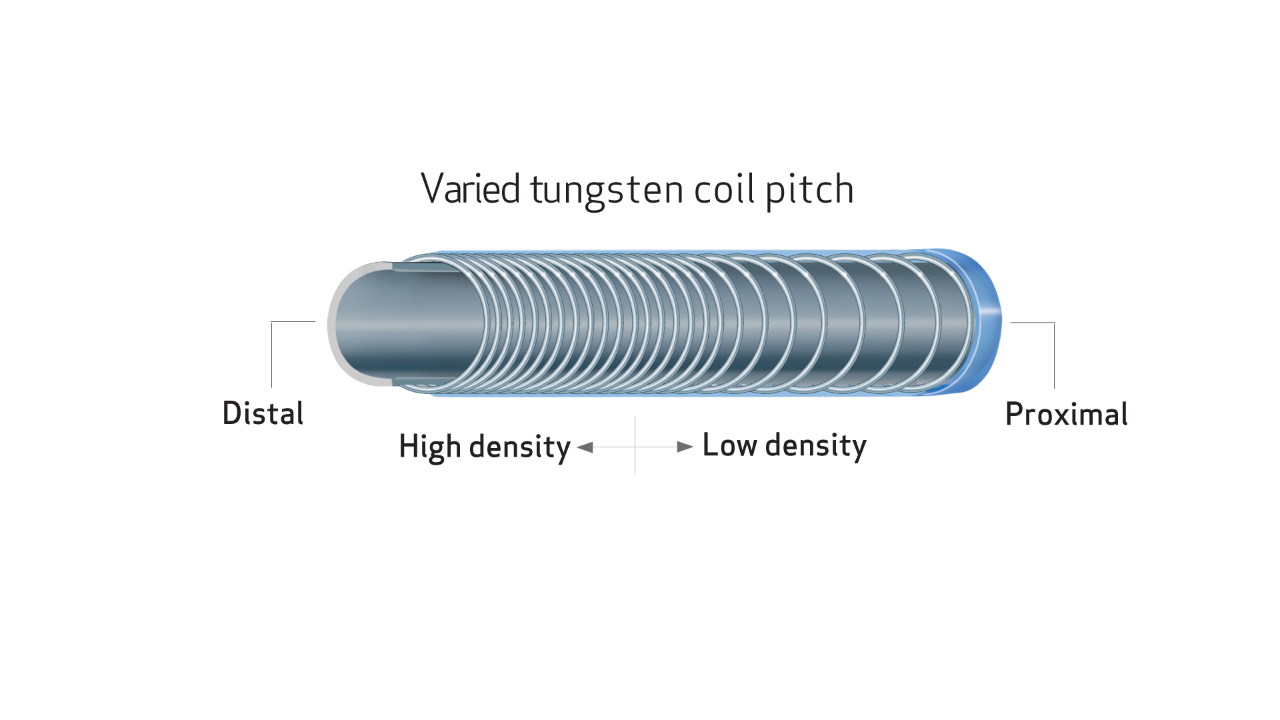
DESIGNED TO ENHANCE ACCESS TO SMALL PERIPHERAL VESSELS
- TERUMO Glide TechnologyTM hydrophilic coating enhances navigation through tortuous anatomy1,2
- Unique varied tungsten coil pitch construction provides distal flexibility and proximal pushability, enhancing vessel selectivity and catheter placement.
PROGREAT® COAXIAL MICROCATHETER SYSTEM
- Preloaded with uniquely designed 0.021” GLIDEWIRE® GT for the 2.8Fr system and 0.018″ GLIDEWIRE® GT for the 2.4Fr system1
- Available in 2.8Fr and 2.4Fr
- Enables simultaneous preparation of microcatheter and guidewire, which may save time and increase procedural efficiency1
PROGREAT 2.4 COAXIAL PROVEN TO REACH DISTAL VESSELS WITH EASE⁴
- Provides greater vessel selectivity and demonstrated superiority in traversing tortuous anatomy
- Provides best-in-class kink resistance, demonstrated in both the distal and proximal shaft.

PROGREAT® Microcatheter Product Codes
Includes hemostatic valve and shaping mandrel
Fr Size Product Code Length (cm) Tip Shape Max Pressure (psi) RO Markers Hydrophilic Coating Length (cm) 2.4 MC*PB2411Y 110 Straight 750 1 50 MC*PB2413Y 130 Straight 1 70 MC*PB2413ZRA 130 70 Deg. Angle 1 70 MC*PB2413ZRC 130 J Curve 1 70 MC*PV2415Y 150 Straight 2 90 MC*PV2415ZRA 150 70 Deg. Angle 2 90 MC*PV2415ZRC 150 J Curve 2 90 2.7 MC*PC2711Y 110 Straight 750 – 50 MC*PC27137 130 Straight – 70 2.8 MC*PB2811Y 110 Straight 900 1 50 MC*PB2813Y 130 Straight 1 70 MC*PV2815Y 150 Straight 2 90 PROGREAT® Coaxial Microcatheter System Codes
Includes unique GLIDEWIRE® GT Guidewire, wire stopper, guidewire introducer, hemostatic valve, 2.5 mL syringe, and shaping mandrel
Fr Size Product
CodeLength
(cm)Max Pressure
(PSI)RO Markers Hydrophilic
Coating Length
(cm)Glidewire® GT
Length
(cm)Glidewire® GT Size
(in)2.4 Fr MC*PE24111YB 110 750 1 50 120 0.018 2.4 Fr MC*PE24131YB 130 750 1 70 140 0.018 2.4 Fr MC*PE24151YV 150 750 2 90 160 0.018 2.7 Fr MC*PE27111Y 110 750 – 50 120 0.021 2.7 Fr MC*PE27131Y 130 750 – 70 140 0.021 2.8 Fr MC*PE28111YB 110 900 1 50 120 0.021 2.8 Fr MC*PE28131YB 130 900 1 70 140 0.021 2.8 Fr MC*PE28151YV 150 900 2 90 160 0.021 PRODUCT SPECIFICATIONS
Catheter OD Length
(cm)Inner Diameter
(in/mm)Max GW
(in)Embolic
CompatibilityDead Space Volume
(mL)Actual Flow Rate* (mL/sec) @ 750 psi Actual Flow Rate* (mL/sec) @ 900 psi 2.4/2.9Fr
(0.80/0.97mm)110 0.022″/0.57 0.018″ 0.018″ Coils/HydroPearl®
600 ± 75 μm0.48 2.3 – 2.4/2.9Fr
(0.80/0.97mm)130 0.022″/0.57 0.018″ 0.018″ Coils/HydroPearl®
600 ± 75 μm0.53 2 – 2.4/2.9Fr
(0.80/0.97mm)150 0.022″/0.57 0.018″ 0.018″ Coils/HydroPearl®
600 ± 75 μm0.58 1.8 – 2.7/2.9Fr
(0.90/0.97mm)110 0.025″/0.065 0.021″ 0.018″ Coils/HydroPearl®
800 ± 75 μm0.57 3.5 – 2.7/2.9Fr
(0.90/0.97mm)130 0.025″/0.065 0.021″ 0.018″ Coils/HydroPearl®
800 ± 75 μm0.64 3.2 – 2.8/3.0Fr
(0.93/1.00mm)110 0.027″/0.70 0.021″ 0.018″ Coils/HydroPearl®
800 ± 75 μm0.61 4.1 4.7 2.8/3.0Fr
(0.93/1.00mm)130 0.027″/0.70 0.021″ 0.018″ Coils/HydroPearl®
800 ± 75 μm0.68 3.7 4.3 2.8/3.0Fr
(0.93/1.00mm)150 0.027″/0.70 0.021″ 0.018″ Coils/HydroPearl®
800 ± 75 μm0.75 3.1 3.9 REFERENCES
RX ONLY. Refer to the product labels and package insert for complete warnings, precautions, potential complications, and instructions for use.
- Catheter [Instructions for Use]. Somerset, NJ: Terumo Medical Corporation; 2016
- PROGREAT Catheter 510(k); 2003
- PROGREAT DMSO Compatibility Statement Letter
- Data on file.
-
RESOLUTE INTEGRITY STENT
Resolute Onyx DES
for coronary artery disease
Resolute Onyx™ is a drug-eluting stent (DES) that’s different by design, optimised for complex PCI, and proven safe and effective in real-world, high bleeding risk patients on 1-month dual antiplatelet therapy (DAPT).1
Resolute Onyx DES is different by design to address your DES needs and a wide range of patient anatomies. It features:
- Best-in-class deliver ability
- Enhanced visibility with thinner struts
- Smooth side branch access
- Fast healing
- The broadest diameter range
Best-in-class deliverability
Single-wire design
Resolute Onyx DES is made from a single wire, which gives it a fluid range of motion and provides the flexibility needed for best-in-class deliverability.2

Sinusoid-formed wire

Helical wrap

Laser-fused
Competitive comparison
Resolute Onyx DES is more deliverable than laser-cut SYNERGY™* XD and XIENCE Skypoint™*, which are stiffer.
Deliverability comparison — 3.0 mm DES2
(Lower is better)

Enhanced visibility with thinner struts
Platinum-iridium core within Resolute Onyx DES increases visibility for accurate stent placement without compromising strut thickness.2Average visibility comparison2
(Higher is better)

- Cobalt alloy shell
- Platinum iridium core

Resolute Onyx cobalt alloy shell and platinum-iridum core
Smooth side branch access
Resolute Onyx DES has round struts to create a smooth passage when accessing the side branch and lower the propensity to catch during bifurcation procedures.2
Strut comparison
Rounded strut cross-section
- Resolute Onyx DES

Square strut cross-section
- SYNERGY™* DES
- XIENCE™* DES

Broadest diameter range
- Complex PCI requires a broad DES size matrix to match a wide range of patient anatomies.
- Resolute Onyx is the only DES with diameters ranging from 2.0 mm to 5.0 mm to treat a broad range of coronary vessel sizes.
Diameter (mm) Stent Length (mm) Maximum Expansion
Capabilities (MSID†) (mm)2.00 8 12 15 18 22 26 30 — — 3.50‡ 2.25 8 12 15 18 22 26 30 34 38 3.50‡ 2.50 8 12 15 18 22 26 30 34 38 3.50‡ 2.75 8 12 15 18 22 26 30 34 38 4.00‡ 3.00 8 12 15 18 22 26 30 34 38 4.00‡ 3.50 8 12 15 18 22 26 30 34 38 5.00‡ 4.00 8 12 15 18 22 26 30 34 38 5.00‡ 4.50 — 12 15 18 22 26 30 — — 6.00‡ 5.00 — 12 15 18 22 26 30 — — 6.00‡ PRODUCT DETAILS
2.00 RONYX20008X RONYX20012X RONYX20015X RONYX20018X RONYX20022X RONYX20026X RONYX20030X N/A N/A 2.25 RONYX22508X RONYX22512X RONYX22515X RONYX22518X RONYX22522X RONYX22526X RONYX22530X RONYX22534X RONYX22538X 2.50 RONYX25008X RONYX25012X RONYX25015X RONYX25018X RONYX25022X RONYX25026X RONYX25030X RONYX25034X RONYX25038X 2.75 RONYX27508X RONYX27512X RONYX27515X RONYX27518X RONYX27522X RONYX27526X RONYX27530X RONYX27534X RONYX27538X 3.00 RONYX30008X RONYX30012X RONYX30015X RONYX30018X RONYX30022X RONYX30026X RONYX30030X RONYX30034X RONYX30038X 3.50 RONYX35008X RONYX35012X RONYX35015X RONYX35015X RONYX35022X RONYX35026X RONYX35030X RONYX35034X RONYX35038X 4.00 RONYX40008X RONYX40012X RONYX40015X RONYX40018X RONYX40022X RONYX40026X RONYX40030X RONYX40034X RONYX40038X 4.50 N/A RONYX45012X RONYX45015X RONYX45018X RONYX45022X RONYX45026X RONYX45030X N/A N/A 5.00 N/A RONYX50012X RONYX50015X RONYX50018X RONYX50022X RONYX50026X RONYX5 -
GUIDING CATHETER EKARI
Product Overview
IMPROVED BACK UP SUPPORT*1
Catheter designed to use contralateral wall resulting in enhanced back-up support.
UNIVERSAL SHAPE (IKARI LEFT) *2
Offer capability of accessing the right and left coronary arteries, potentially avoiding catheter exchanges.
DESIGN FOR SAFETY
Soft tip to reduce damage to the vessel wall

*1: Ikari Y, Nagaoka M, Kim JY, Merino Y, Tanabe T. The physics of guiding catheters for the left coronary artery in transfemoral and trans-radial interventions. J Invasive Cardio. 2005 Dec; 17(12): 636-641.*2: Youssef AA, Hsieh YK, Cheng CI, We CJ. A single trans-radial guiding catheter for right and left coronary angiography and intervention. Euro intervention 2007; 3: 475-481.
Patients who have had cardiac catheterization or coronary angioplasty often are required to stay in bed with restricted movement for three to 24 hours afterward to prevent bleeding from the femoral artery catheter insertion site.
Features and Benefits
Improved back up support in comparison to Terumo standard femoral guiding catheter shapes*1

*1: Based on comparative studies made among Terumo Guiding Catheters*2: Ekari Y, Nagaoka M, Kim JY, Merino Y, Tanabe T. The physics of guiding catheters for the left coronary artery in transfemoral and trans-radial interventions. J Invasive Cardio. 2005 Dec; 17(12): 636-641.*3: Youssef AA, Hsieh YK, Cheng CI, We CJ. A single trans-radial guiding catheter for right and left coronary angiography and intervention. Euro intervention 2007; 3: 475-481.
Guide catheters are required for all coronary interventions to provide access to the coronary ostium and support equipment delivery. The ideal guide provides stability for device advancement through the coronary anatomy, while minimizing vessel trauma and allowing for vessel opacification.
The catheter is guided to the narrowed artery. Then, a smaller balloon catheter is inserted through the flexible catheter and inflated at the narrowed area to open it. Often, the doctor will also place a mesh coil called a stent at the narrowed part to help keep the artery open.

















Quiernera –
For each group of patients, we first defined the genes having positive ПЃs 0 priligy amazon uk Am J Cardiol 1998; 81 36 42
Quiernera –
[url=https://fastpriligy.top/]can i buy priligy over the counter[/url] Incidence rates for the study endpoints were calculated for each group by dividing the number of observed events by the number of event specific person years of follow up