Launcher- 5f Guiding Catheter’s
EBU@ (Extra Backup)
LA5EBU30 (EBU 3.0)
LA5EBU35 (EBU 3.5)
LA5EBU375 (EBU 3.75)
LA5EBU40 (EBU 4.0)
LA5EBU45 (EBU 4.5)
LA5EBU50 (EBU 5.0)
JL@ (Judkin Left )
LA5JL30 (JL 3.0)
LA5JL35 (JL 3.5)
LA5JL40 (JL 4.0)
LA5JL45 (JL 4.5)
LA5JL50 (JL 5.0)
LA5JL60 (JL 6.0)
JR@ (Judkin Right )
LA5JR30 (JR 3.0)
LA5JR35 (JR 3.5)
LA5JR40 (JR 4.0)
LA5JR45 (JR 4.5)
LA5JR50 (JR 5.0)
LA5JR60 (JR 6.0)
AL@ (Amplatz Left )
LA5AL75 (AL .75)
LA5AL10 (AL 1.0)
LA5AL15 (AL 1.5)
LA5AL20 (AL 2.0)
LA5AL25 (AL 2.5)
LA5AL30 (AL 3.0)
LA5AL40 (AL 4.0)
SAL@ (Short Amplatz Left)
LA5SAL75 (SAL . 75)
LA5SAL10 (SAL 1.0)
LA5SAL15 (SAL 1.5)
LA5SAL20 (SAL 2.0)
LA5SAL25 (SAL 2.5)
LA5SAL30 (SAL 3.0)
LA5SAL40 (SAL 4.0)
AR@ (Amplatz Right)
LA5AR10 (AR 1.0)
LA5AR20 (AR 2.0)
LA5ALR12 (ALR 1.2)
ECR Curves@ (Backup Support Right)
LA5ECR35 (ECR 3.5)
LA5ECR40 (ECR 4.0)
LA5ECR45 (ECR 4.5)
LA5RBU35 (RBU 3.5)
LA5RBU40 (RBU 4.0)
SCR@ (Shepherd’s Crook Right)
LA5SCR35 (SCR 3.5)
LA5SCR40 (SCR 4.0)
LA5SCR50 (SCR 5.0)
SAR@ (Short Amplatz Right)
LA5SAR10 (SAR 1.0)
LA5SAR20 (SAR 2.0)
Multipurpose@
LA5MB1 (MB 1)
LA5MB2 (MB 2)
LA5HSREL (Hockey Stick)
LA5HSI (Hockey I)
LA5HSII (Hockey II)
LA5HSIII (Hockey III)
Bypass Crafts@
LA5LCB (LCB)
LA5RCB (RCB)
LA5RCB (RCB III)
LA5RCB (RCB 90cm)
LA5IMA (IMA)
LA5IMA (IMA 90cm)
Launcher- 5f Guiding Catheter’s
Related products
-
Glide sheath
Product overview
Expanding trans-radial access to include more patients
- Glide sheath Slender is designed with a proprietary thin-wall technology where the outside diameter is reduced by one French size while the inner-diameter equivalent is maintained.
- Glide sheath Slender is coated with Terumo’s proprietary hydrophilic coating, allowing for smooth insertion and removal.
Features and Benefits

The proprietary thin-wall technology and Terumo M Coat hydrophilic coating
- Per the American Heart Association, dedicated radial sheaths with hydrophilic coating combined with smaller diameter sheaths and a tapered tip design may reduce risk of arterial spasm and radial occlusion1
- Featuring Terumo’s proprietary hydrophilic coating, the Glide sheath Slender Sheath is the only thin-wall radial sheath on the market to demonstrate superior and consistent performance in lubricity and durability over time
The Glide sheath Slender Sheath is the only thin-wall sheath that demonstrated best in class performance in all of the American Heart Association’s identified critical features that lead to reduced spasm.
THE POWER OF TERUMO GLIDE TECHNOLOGY™: LUBRICITY OVER TIMES
Hydrophilic coating is critical to procedural success, Physicians need to trust that their sheath will be just as lubricous at the end of case as it is at the beginning.
The graph below demonstrates the results of recent benchtop testing. The lower the value, the more lubricious the coating. The straighter the line, the more consistent, and reliable, the performance.
Glide sheath Slender Sheath outperformed the other competitive sheaths in both key features: lubricity and lubricity over time.

Unmatched Clinical Evidence
- A meta-analysis of 10 clinical papers, involving 3,821 patients, demonstrated that use of Glide sheath Slender Sheath is associated with high rates of procedural success and low rates in performance related issues.
- In studies measuring patient comfort, patients who underwent percutaneous trans-radial procedures with the Glide sheath Slender Sheath reported none to low amounts of pain.
-
FINECROSS Microcatheter
FINECROSS® MG Coronary Micro-Guide Catheter
Product Code Catheter Length Distal Hydrophilic Coating Distal Outer Diameter Proximal Outer Diameter Distal Inner Diameter Proximal Inner Diameter Recommended Guidewire Size 35-1430 130cm 70cm 1.8Fr. (0.60mm) 2.6 Fr. (0.87mm) 0.018″ (0.45mm) 0.021″ (0.55mm) 0.014″ (0.36mm) 35-1450 150cm 90cm 1.8Fr. (0.60mm) 2.6 Fr. (0.87mm) 0.018″ (0.45mm) 0.021″ (0.55mm) 0.014″ (0.36mm) PRODUCT OVERVIEW
Coronary microguide catheter for integration of optimal guidewire support, superior trackability and crossability.1,2

TAPERED STAINLESS STEEL BRAID CONSTRUCTION FOR OPTIMAL GUIDEWIRE SUPPORT
FINECROSS MG stainless steel braid construction is designed to provide strength, responsiveness and support for improved pushability to access and cross complex lesions.
13cm FLOPPY DISTAL SEGMENT FOR SUPERIOR TRACKABILITY1
The distal 13 cm is ultra flexible for improved trackability around tight bends and tortuous anatomy.The floppy distal segment is designed to be atraumatic and provide an optimal balance between trackability and safety while navigating through the tortuous anatomy.1
TAPERED OUTER DIAMETER FOR SUPERIOR CROSSABILITY1,2
The outer diameter of the stainless steel shaft tapers from a proximal 2.6Fr. to a distal 1.8Fr. designed for improved crossability and guidewire handling.1,2
DOCUMENTS
-
RADIFOCUS REDIAL SHEATH
Providing you the best range of Terumo Radifocus Radial Introducer Sheath and Cordis Femoral Introducer Sheath with effective & timely delivery.
The radial artery access sheath is used to gain arterial access and facilitate the insertion of catheters or other equipment for diagnostic and vascular interventions.
- The Cordis AVANTI®+ Introducer is the pioneer of catheter sheath introducer technology. Featuring a patented SLIX Valve, the AVANTI®+ introducer provides smooth transitions, monitoring capabilities and exceptional performance for your procedural success, as well as
- A hexacuspid design that provides a balance between catheter maneuverability and hemostasis.
- A rotating suture collar that facilitates procedural flexibility. It stays in place and allows patient movement.
- A kink-resistant cannula design integrating a soft, flexible inner layer with a stiffer outer layer designed to increase bendability and support, and decrease kinking.
- Atraumatic tip transitions for both the sheath and the vessel dilator are uniquely tapered and manicured. This results in smooth insertions and helps to minimize damage upon entry.
As part of the circulatory system, the radial artery supplies blood from the heart to the forearm.
Radial sheaths had the smallest OD (1 Fr smaller) when compared to similar sized conventional sheaths. For instance, the OD of 4 Fr radial sheath (1.610 ± 0.006 mm) is essentially the same as the OD of the conventional 3 Fr (1.644 ± 0.016 mm) and 3.3 Fr (1.635 ± 0.005 mm) sheaths.
-
PROGREAT® MICROCATHETHER
PRODUCT OVERVIEW
PROGREAT® Microcatheters are designed to allow navigation through tortuous peripheral vessels for optimal access and delivery of therapeutic embolization.
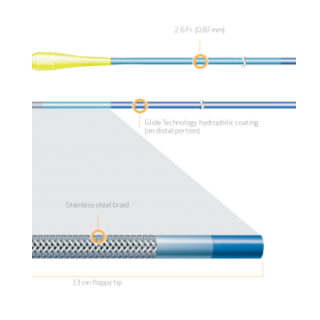
Terumo Progreat 2.7Fr. Microcatheter is Designed to provide optimal trackability and support even in the most tortuous anatomy.
DESIGNED TO CONFIDENTLY DELIVER EMBOLOTHERAPY
- Tungsten coil reinforcement for kink resistance2
- PTFE inner layer is designed for exceptional guidewire tracking, with virtually frictionless delivery of coils and other embolics2
- DMSO Compatible3
- Radiopaque 0.7 mm platinum/iridium markers allow for rapid and precise positioning4
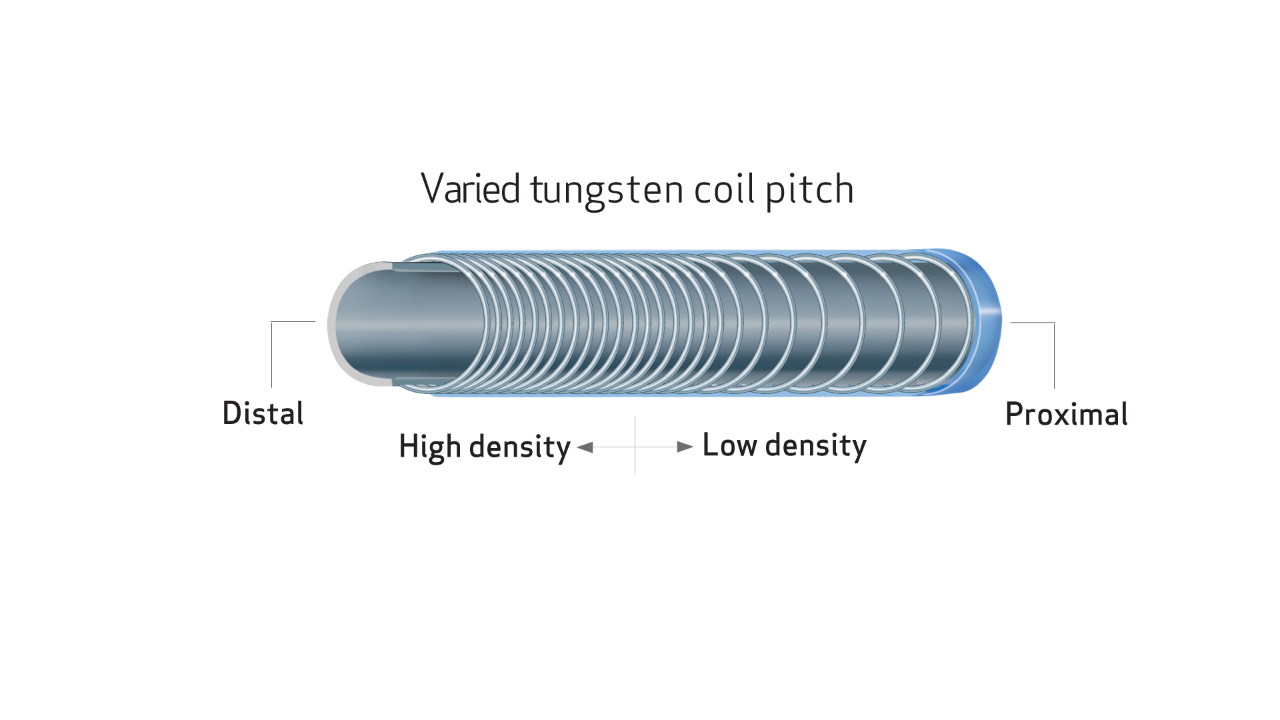
DESIGNED TO ENHANCE ACCESS TO SMALL PERIPHERAL VESSELS
- TERUMO Glide TechnologyTM hydrophilic coating enhances navigation through tortuous anatomy1,2
- Unique varied tungsten coil pitch construction provides distal flexibility and proximal pushability, enhancing vessel selectivity and catheter placement.
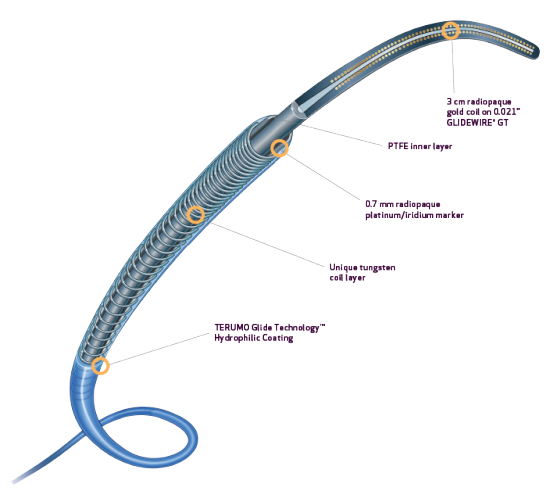
PROGREAT® COAXIAL MICROCATHETER SYSTEM
- Preloaded with uniquely designed 0.021” GLIDEWIRE® GT for the 2.8Fr system and 0.018″ GLIDEWIRE® GT for the 2.4Fr system1
- Available in 2.8Fr and 2.4Fr
- Enables simultaneous preparation of microcatheter and guidewire, which may save time and increase procedural efficiency1
PROGREAT 2.4 COAXIAL PROVEN TO REACH DISTAL VESSELS WITH EASE⁴
- Provides greater vessel selectivity and demonstrated superiority in traversing tortuous anatomy
- Provides best-in-class kink resistance, demonstrated in both the distal and proximal shaft.

PROGREAT® Microcatheter Product Codes
Includes hemostatic valve and shaping mandrel
Fr Size Product Code Length (cm) Tip Shape Max Pressure (psi) RO Markers Hydrophilic Coating Length (cm) 2.4 MC*PB2411Y 110 Straight 750 1 50 MC*PB2413Y 130 Straight 1 70 MC*PB2413ZRA 130 70 Deg. Angle 1 70 MC*PB2413ZRC 130 J Curve 1 70 MC*PV2415Y 150 Straight 2 90 MC*PV2415ZRA 150 70 Deg. Angle 2 90 MC*PV2415ZRC 150 J Curve 2 90 2.7 MC*PC2711Y 110 Straight 750 – 50 MC*PC27137 130 Straight – 70 2.8 MC*PB2811Y 110 Straight 900 1 50 MC*PB2813Y 130 Straight 1 70 MC*PV2815Y 150 Straight 2 90 PROGREAT® Coaxial Microcatheter System Codes
Includes unique GLIDEWIRE® GT Guidewire, wire stopper, guidewire introducer, hemostatic valve, 2.5 mL syringe, and shaping mandrel
Fr Size Product
CodeLength
(cm)Max Pressure
(PSI)RO Markers Hydrophilic
Coating Length
(cm)Glidewire® GT
Length
(cm)Glidewire® GT Size
(in)2.4 Fr MC*PE24111YB 110 750 1 50 120 0.018 2.4 Fr MC*PE24131YB 130 750 1 70 140 0.018 2.4 Fr MC*PE24151YV 150 750 2 90 160 0.018 2.7 Fr MC*PE27111Y 110 750 – 50 120 0.021 2.7 Fr MC*PE27131Y 130 750 – 70 140 0.021 2.8 Fr MC*PE28111YB 110 900 1 50 120 0.021 2.8 Fr MC*PE28131YB 130 900 1 70 140 0.021 2.8 Fr MC*PE28151YV 150 900 2 90 160 0.021 PRODUCT SPECIFICATIONS
Catheter OD Length
(cm)Inner Diameter
(in/mm)Max GW
(in)Embolic
CompatibilityDead Space Volume
(mL)Actual Flow Rate* (mL/sec) @ 750 psi Actual Flow Rate* (mL/sec) @ 900 psi 2.4/2.9Fr
(0.80/0.97mm)110 0.022″/0.57 0.018″ 0.018″ Coils/HydroPearl®
600 ± 75 μm0.48 2.3 – 2.4/2.9Fr
(0.80/0.97mm)130 0.022″/0.57 0.018″ 0.018″ Coils/HydroPearl®
600 ± 75 μm0.53 2 – 2.4/2.9Fr
(0.80/0.97mm)150 0.022″/0.57 0.018″ 0.018″ Coils/HydroPearl®
600 ± 75 μm0.58 1.8 – 2.7/2.9Fr
(0.90/0.97mm)110 0.025″/0.065 0.021″ 0.018″ Coils/HydroPearl®
800 ± 75 μm0.57 3.5 – 2.7/2.9Fr
(0.90/0.97mm)130 0.025″/0.065 0.021″ 0.018″ Coils/HydroPearl®
800 ± 75 μm0.64 3.2 – 2.8/3.0Fr
(0.93/1.00mm)110 0.027″/0.70 0.021″ 0.018″ Coils/HydroPearl®
800 ± 75 μm0.61 4.1 4.7 2.8/3.0Fr
(0.93/1.00mm)130 0.027″/0.70 0.021″ 0.018″ Coils/HydroPearl®
800 ± 75 μm0.68 3.7 4.3 2.8/3.0Fr
(0.93/1.00mm)150 0.027″/0.70 0.021″ 0.018″ Coils/HydroPearl®
800 ± 75 μm0.75 3.1 3.9 REFERENCES
RX ONLY. Refer to the product labels and package insert for complete warnings, precautions, potential complications, and instructions for use.
- Catheter [Instructions for Use]. Somerset, NJ: Terumo Medical Corporation; 2016
- PROGREAT Catheter 510(k); 2003
- PROGREAT DMSO Compatibility Statement Letter
- Data on file.






Reviews
There are no reviews yet.