Launcher- 7f Guiding Catheter’s
EBU@ (Extra Backup)
LA7EBU30 (EBU 3.0)
LA7EBU35 (EBU 3.5)
LA7EBU375 (EBU 3.75)
LA7EBU40 (EBU 4.0)
LA7EBU45 (EBU 4.5)
LA7EBU50 (EBU 5.0)
JL@ (Judkin Left )
LA7JL30 (JL 3.0)
LA7JL35 (JL 3.5)
LA7JL40 (JL 4.0)
LA7JL45 (JL 4.5)
LA7JL50 (JL 5.0)
LA7JL60 (JL 6.0)
JR@ (Judkin Right )
LA7JR30 (JR 3.0)
LA7JR35 (JR 3.5)
LA7JR40 (JR 4.0)
LA7JR45 (JR 4.5)
LA7JR50 (JR 5.0)
LA7JR60 (JR 6.0)
AL@ (Amplatz Left )
LA7AL75 (AL .75)
LA7AL10 (AL 1.0)
LA7AL15 (AL 1.5)
LA7AL20 (AL 2.0)
LA7AL25 (AL 2.5)
LA7AL30 (AL 3.0)
LA7AL40 (AL 4.0)
SAL@ (Short Amplatz Left)
LA7SAL75 (SAL . 75)
LA7SAL10 (SAL 1.0)
LA7SAL15 (SAL 1.5)
LA7SAL20 (SAL 2.0)
LA7SAL25 (SAL 2.5)
LA7SAL30 (SAL 3.0)
LA7SAL40 (SAL 4.0)
AR@ (Amplatz Right)
LA7AR10 (AR 1.0)
LA7AR20 (AR 2.0)
LA7ALR12 (ALR 1.2)
ECR Curves@ (Backup Support Right)
LA7ECR35 (ECR 3.5)
LA7ECR40 (ECR 4.0)
LA7ECR45 (ECR 4.5)
LA7RBU35 (RBU 3.5)
LA7RBU40 (RBU 4.0)
SCR@ (Shepherd’s Crook Right)
LA7SCR35 (SCR 3.5)
LA7SCR40 (SCR 4.0)
LA7SCR50 (SCR 5.0)
SAR@ (Short Amplatz Right)
LA7SAR10 (SAR 1.0)
LA7SAR20 (SAR 2.0)
Multipurpose@
LA7MB1 (MB 1)
LA7MB2 (MB 2)
LA7HSREL (Hockey Stick)
LA7HSI (Hockey I)
LA7HSII (Hockey II)
LA7HSIII (Hockey III)
Bypass Crafts@
LA7LCB (LCB)
LA7RCB (RCB)
LA7RCB (RCB III)
LA7RCB (RCB 90cm)
LA7IMA (IMA)
LA7IMA (IMA 90cm)
Launcher- 7f Guiding Catheter’s
Related products
-
GUIDING CATHETER EKARI
Product Overview
IMPROVED BACK UP SUPPORT*1
Catheter designed to use contralateral wall resulting in enhanced back-up support.
UNIVERSAL SHAPE (IKARI LEFT) *2
Offer capability of accessing the right and left coronary arteries, potentially avoiding catheter exchanges.
DESIGN FOR SAFETY
Soft tip to reduce damage to the vessel wall

*1: Ikari Y, Nagaoka M, Kim JY, Merino Y, Tanabe T. The physics of guiding catheters for the left coronary artery in transfemoral and trans-radial interventions. J Invasive Cardio. 2005 Dec; 17(12): 636-641.*2: Youssef AA, Hsieh YK, Cheng CI, We CJ. A single trans-radial guiding catheter for right and left coronary angiography and intervention. Euro intervention 2007; 3: 475-481.
Patients who have had cardiac catheterization or coronary angioplasty often are required to stay in bed with restricted movement for three to 24 hours afterward to prevent bleeding from the femoral artery catheter insertion site.
Features and Benefits
Improved back up support in comparison to Terumo standard femoral guiding catheter shapes*1

*1: Based on comparative studies made among Terumo Guiding Catheters*2: Ekari Y, Nagaoka M, Kim JY, Merino Y, Tanabe T. The physics of guiding catheters for the left coronary artery in transfemoral and trans-radial interventions. J Invasive Cardio. 2005 Dec; 17(12): 636-641.*3: Youssef AA, Hsieh YK, Cheng CI, We CJ. A single trans-radial guiding catheter for right and left coronary angiography and intervention. Euro intervention 2007; 3: 475-481.
Guide catheters are required for all coronary interventions to provide access to the coronary ostium and support equipment delivery. The ideal guide provides stability for device advancement through the coronary anatomy, while minimizing vessel trauma and allowing for vessel opacification.
The catheter is guided to the narrowed artery. Then, a smaller balloon catheter is inserted through the flexible catheter and inflated at the narrowed area to open it. Often, the doctor will also place a mesh coil called a stent at the narrowed part to help keep the artery open.
-
PROGREAT® MICROCATHETHER
PRODUCT OVERVIEW
PROGREAT® Microcatheters are designed to allow navigation through tortuous peripheral vessels for optimal access and delivery of therapeutic embolization.
Terumo Progreat 2.7Fr. Microcatheter is Designed to provide optimal trackability and support even in the most tortuous anatomy.
DESIGNED TO CONFIDENTLY DELIVER EMBOLOTHERAPY
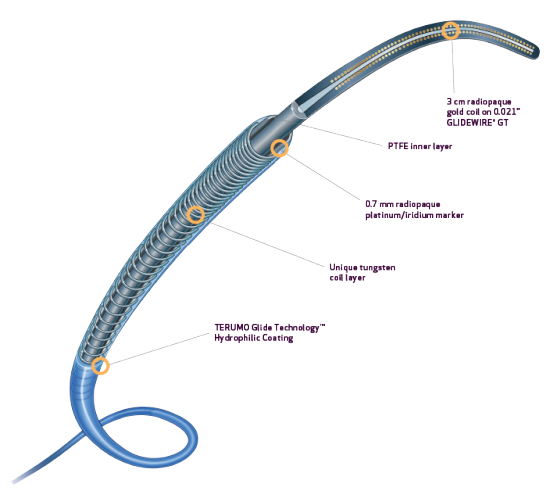
- Tungsten coil reinforcement for kink resistance2
- PTFE inner layer is designed for exceptional guidewire tracking, with virtually frictionless delivery of coils and other embolics2
- DMSO Compatible3
- Radiopaque 0.7 mm platinum/iridium markers allow for rapid and precise positioning4
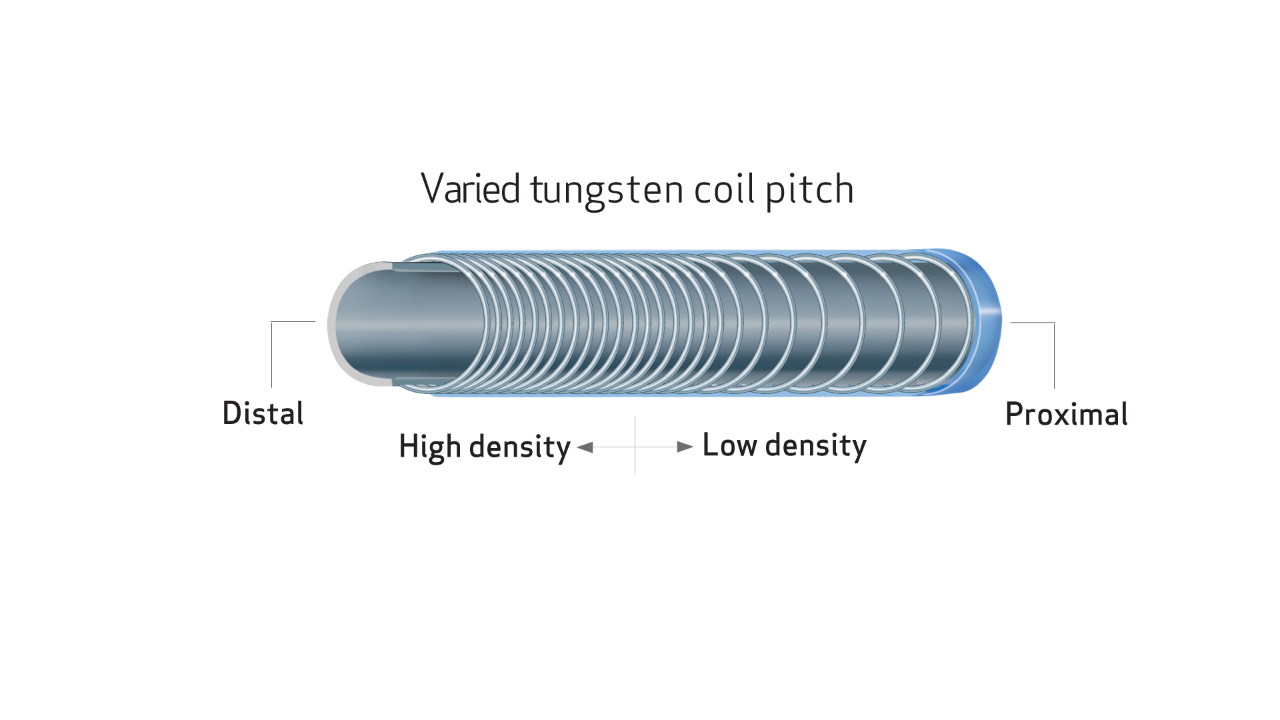
DESIGNED TO ENHANCE ACCESS TO SMALL PERIPHERAL VESSELS
- TERUMO Glide TechnologyTM hydrophilic coating enhances navigation through tortuous anatomy1,2
- Unique varied tungsten coil pitch construction provides distal flexibility and proximal pushability, enhancing vessel selectivity and catheter placement.
PROGREAT® COAXIAL MICROCATHETER SYSTEM
- Preloaded with uniquely designed 0.021” GLIDEWIRE® GT for the 2.8Fr system and 0.018″ GLIDEWIRE® GT for the 2.4Fr system1
- Available in 2.8Fr and 2.4Fr
- Enables simultaneous preparation of microcatheter and guidewire, which may save time and increase procedural efficiency1
PROGREAT 2.4 COAXIAL PROVEN TO REACH DISTAL VESSELS WITH EASE⁴
- Provides greater vessel selectivity and demonstrated superiority in traversing tortuous anatomy
- Provides best-in-class kink resistance, demonstrated in both the distal and proximal shaft.

PROGREAT® Microcatheter Product Codes
Includes hemostatic valve and shaping mandrel
Fr Size Product Code Length (cm) Tip Shape Max Pressure (psi) RO Markers Hydrophilic Coating Length (cm) 2.4 MC*PB2411Y 110 Straight 750 1 50 MC*PB2413Y 130 Straight 1 70 MC*PB2413ZRA 130 70 Deg. Angle 1 70 MC*PB2413ZRC 130 J Curve 1 70 MC*PV2415Y 150 Straight 2 90 MC*PV2415ZRA 150 70 Deg. Angle 2 90 MC*PV2415ZRC 150 J Curve 2 90 2.7 MC*PC2711Y 110 Straight 750 – 50 MC*PC27137 130 Straight – 70 2.8 MC*PB2811Y 110 Straight 900 1 50 MC*PB2813Y 130 Straight 1 70 MC*PV2815Y 150 Straight 2 90 PROGREAT® Coaxial Microcatheter System Codes
Includes unique GLIDEWIRE® GT Guidewire, wire stopper, guidewire introducer, hemostatic valve, 2.5 mL syringe, and shaping mandrel
Fr Size Product
CodeLength
(cm)Max Pressure
(PSI)RO Markers Hydrophilic
Coating Length
(cm)Glidewire® GT
Length
(cm)Glidewire® GT Size
(in)2.4 Fr MC*PE24111YB 110 750 1 50 120 0.018 2.4 Fr MC*PE24131YB 130 750 1 70 140 0.018 2.4 Fr MC*PE24151YV 150 750 2 90 160 0.018 2.7 Fr MC*PE27111Y 110 750 – 50 120 0.021 2.7 Fr MC*PE27131Y 130 750 – 70 140 0.021 2.8 Fr MC*PE28111YB 110 900 1 50 120 0.021 2.8 Fr MC*PE28131YB 130 900 1 70 140 0.021 2.8 Fr MC*PE28151YV 150 900 2 90 160 0.021 PRODUCT SPECIFICATIONS
Catheter OD Length
(cm)Inner Diameter
(in/mm)Max GW
(in)Embolic
CompatibilityDead Space Volume
(mL)Actual Flow Rate* (mL/sec) @ 750 psi Actual Flow Rate* (mL/sec) @ 900 psi 2.4/2.9Fr
(0.80/0.97mm)110 0.022″/0.57 0.018″ 0.018″ Coils/HydroPearl®
600 ± 75 μm0.48 2.3 – 2.4/2.9Fr
(0.80/0.97mm)130 0.022″/0.57 0.018″ 0.018″ Coils/HydroPearl®
600 ± 75 μm0.53 2 – 2.4/2.9Fr
(0.80/0.97mm)150 0.022″/0.57 0.018″ 0.018″ Coils/HydroPearl®
600 ± 75 μm0.58 1.8 – 2.7/2.9Fr
(0.90/0.97mm)110 0.025″/0.065 0.021″ 0.018″ Coils/HydroPearl®
800 ± 75 μm0.57 3.5 – 2.7/2.9Fr
(0.90/0.97mm)130 0.025″/0.065 0.021″ 0.018″ Coils/HydroPearl®
800 ± 75 μm0.64 3.2 – 2.8/3.0Fr
(0.93/1.00mm)110 0.027″/0.70 0.021″ 0.018″ Coils/HydroPearl®
800 ± 75 μm0.61 4.1 4.7 2.8/3.0Fr
(0.93/1.00mm)130 0.027″/0.70 0.021″ 0.018″ Coils/HydroPearl®
800 ± 75 μm0.68 3.7 4.3 2.8/3.0Fr
(0.93/1.00mm)150 0.027″/0.70 0.021″ 0.018″ Coils/HydroPearl®
800 ± 75 μm0.75 3.1 3.9 REFERENCES
RX ONLY. Refer to the product labels and package insert for complete warnings, precautions, potential complications, and instructions for use.
- Catheter [Instructions for Use]. Somerset, NJ: Terumo Medical Corporation; 2016
- PROGREAT Catheter 510(k); 2003
- PROGREAT DMSO Compatibility Statement Letter
- Data on file.
-
FINECROSS Microcatheter
FINECROSS® MG Coronary Micro-Guide Catheter
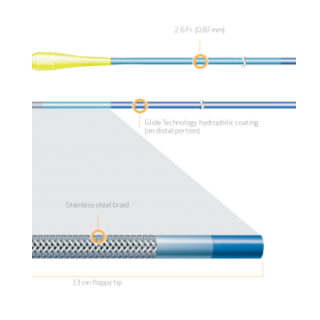
Product Code Catheter Length Distal Hydrophilic Coating Distal Outer Diameter Proximal Outer Diameter Distal Inner Diameter Proximal Inner Diameter Recommended Guidewire Size 35-1430 130cm 70cm 1.8Fr. (0.60mm) 2.6 Fr. (0.87mm) 0.018″ (0.45mm) 0.021″ (0.55mm) 0.014″ (0.36mm) 35-1450 150cm 90cm 1.8Fr. (0.60mm) 2.6 Fr. (0.87mm) 0.018″ (0.45mm) 0.021″ (0.55mm) 0.014″ (0.36mm) PRODUCT OVERVIEW
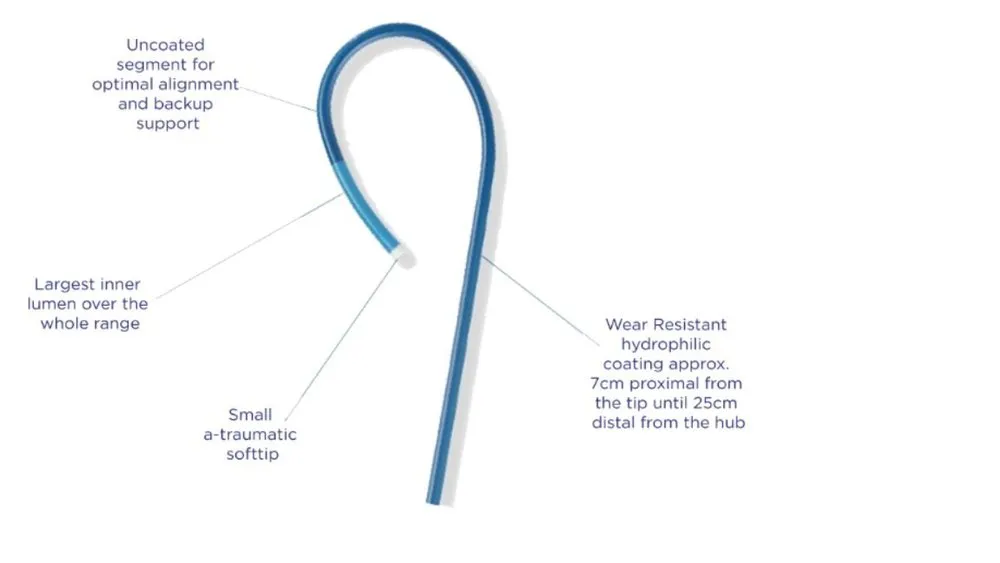
Coronary microguide catheter for integration of optimal guidewire support, superior trackability and crossability.1,2

TAPERED STAINLESS STEEL BRAID CONSTRUCTION FOR OPTIMAL GUIDEWIRE SUPPORT
FINECROSS MG stainless steel braid construction is designed to provide strength, responsiveness and support for improved pushability to access and cross complex lesions.
13cm FLOPPY DISTAL SEGMENT FOR SUPERIOR TRACKABILITY1
The distal 13 cm is ultra flexible for improved trackability around tight bends and tortuous anatomy.The floppy distal segment is designed to be atraumatic and provide an optimal balance between trackability and safety while navigating through the tortuous anatomy.1
TAPERED OUTER DIAMETER FOR SUPERIOR CROSSABILITY1,2
The outer diameter of the stainless steel shaft tapers from a proximal 2.6Fr. to a distal 1.8Fr. designed for improved crossability and guidewire handling.1,2
DOCUMENTS
-
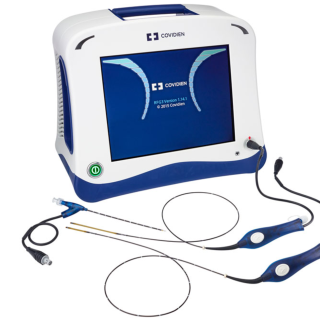
CLOSUREFAST
ClosureFast™
procedureMinimally invasive thermal
treatment for vein disease
and varicose veins.
The ClosureFast procedure uses radiofrequency energy or heat to close the diseased vein, which redirects blood flow to healthy veins, relieving symptoms.Before the procedure
You will have an ultrasound imaging exam of the leg being treated. This exam is important for assessing the diseased superficial vein and planning the procedure.
During the procedure
Your doctor will discuss the procedure with you. Here is a brief summary of what to expect:
- The ClosureFast procedure is performed on an outpatient basis. Your doctor will perform the ClosureFast procedure. Using an ultrasound, your doctor will position a catheter in the diseased vein through a small opening in the skin.
- The small catheter delivers heat to the vein wall, causing it to shrink and seal the vein. Once the diseased vein is closed, blood will reroute itself to other healthy veins.
After the procedure
- You will be taken to the recovery area to rest.
- Following the procedure, your doctor will apply a simple bandage over the insertion site and will give you compression stockings to aid in the healing process. Your doctor may encourage you to walk and to refrain from extended standing and strenuous activities for a period of time. The average patient typically resumes normal activities within a few days.1,2
- Your doctor will recommend follow-up care as needed.
-
Benefits of the ClosureFast procedure
- Relief of symptoms after two days, with a noticeable improvement in one to two week
- Minimally invasive outpatient procedure
- Less pain and bruising than laser treatment
- Faster recovery than laser treatment
- Proven results with positive patient experience
- Rapid recovery — on average, patients resume normal activities within a few days
- Lasting results — the only radiofrequency energy procedure with published long-term clinical data demonstrating safety and efficacy, with a 91.9% closure rate at five years.4
Adverse events can include nerve injury, hematoma, phlebitis, thrombosis, and/or pulmonary embolism.
Type of therapy
Thermal shrinkage of the vein wall with radiofrequency energy or heatInpatient or outpatient procedure
Outpatient procedureOne- versus two-leg treatment
Typically, each leg will be treated in separate appointmentsNeedle sticks required
One needle stick for vein access. Typically, 5 to 10 needle sticks for anesthesia.Typical post-treatment recovery
Healing of vein access site, anesthetic needle stick sites, and also healing of ablated vein sectionCompression hose
Required for approximately one weekProcedure success rate
91.9% after five years4
ClosureFast procedure FAQ
Is the ClosureFast procedure painful?
Most patients report feeling little, if any, pain during the ClosureFast procedure.3 Your doctor will give you a local or regional anesthetic to numb the treatment area.
When can I return to normal activity?
Patients treated with the ClosureFast procedure may resume normal activity more quickly than patients who undergo surgical vein stripping or laser ablation. With the ClosureFast procedure, the average patient typically resumes normal activity within a few days.2 For a few weeks following the treatment, your vein specialist may recommend a regular walking regimen and suggest you refrain from strenuous activities (heavy lifting, for example) or prolonged periods of standing.
When will my symptoms improve?
Most patients report relief of symptoms after two days, with a noticeable improvement in one to two weeks.
Is there any scarring, bruising, or swelling after the procedure?
Most patients report limited to no scarring, bruising, or swelling following the ClosureFast procedure.3
How is the ClosureFast procedure different from endovenous lasers?
Both Closure Fast and lasers use thermal technology to deliver heat into the diseased vein, but they each use a different method of delivery. Despite their similarity in using heat, a 2009 study showed that the ClosureFast procedure is associated with lower rates of pain, bruising, and complications and a faster improvement in patients’ quality of life when compared to 980 nm laser ablation.4
How is the Closure Fast procedure different from vein stripping?
During vein stripping, incisions are made in the groin and calf, and a tool is threaded through the diseased vein to pull the vein out of the leg. With the ClosureFast procedure, only one small incision is made at the insertion site and the vein is then treated and left in place. ClosureFast is a minimally invasive approach that reduces the likelihood of pain and bruising, associated with vein stripping surgery.1
Is the ClosureFast procedure covered by insurance?
Many patients have access to the ClosureFast procedure through their insurance coverage plan. Insurance companies detail access to the ClosureFast procedure and other radiofrequency ablation procedures in coverage policies for varicose veins or chronic venous insufficiency. As with all healthcare procedures, you may also have some cost associated with receiving treatment, such as a copay or coinsurance. Please reach out to your insurance company to discuss your specific plan coverage and potential costs prior to seeking treatment







Reviews
There are no reviews yet.