Heartrail® II – PTCA Guiding Catheter
Heartrail® II – PTCA Guiding Catheter
Heartrail II is developed to maximize your back-up force when using right and left Ikari curves during transradial interventions and through its innovative 5-in-6 system.
Its high-strength wire braiding provides exceptional kink-resistance in tortuous blood vessels, while assuring superior torque response. It also has gentle tip and powerful shaft performance.
General Specifications
| Shaft inner layer material | Polytetrafluoroethylene – PTFE |
|---|---|
| Braiding | High strength flat wire mesh pattern |
| Inner diameter | 5 Fr – 0.059″ (1.50 mm) 6 Fr – 0.071″ (1.80 mm) 7 Fr – 0.081″ (2.06 mm) |
| Usable length | 100 cm 120 cm – Heartrail 5 in 6 system |
| Sizes Available | Item Code |
|---|---|
| AL Short Tip 6F | TCO06GC-F6AL1SON |
| AL0.75/6F | TCO06GC-F6AL0G0N |
| AL1.0/6F | TCO06GC-F6AL010N |
| BL3.0/6F | TCO06GC-F6BL300N |
| BL3.5/6F | TCO06GC-F6BL350N |
| BL3.5/7F | TCO06GC-F7BL350N |
| BL4.0/6F | TCO06GC-F6BL400N |
| IKARI IL 3.5/5F | TCO06GC-F5IL350N |
| IKARI IL 3.5/6F | TCO06GC-F6IL350N |
| IKARI IL 4.0/6F | TCO06GC-F6IL400N |
| IKARI RIGHT 1.5/6F | TCO06GC-F6IR150N |
| Sizes Available | Item Code |
|---|---|
| JL3.5/5F | TCO06GC-F5JL350N |
| JL3.5/6F | TCO06GC-F6JL350N |
| JL4.0/6F | TCO06GC-F6JL400N |
| JR3.5/5F | TCO06GC-F5JR350N |
| JR3.5/6F | TCO06GC-F6JR350N |
| JR4.0/5F | TCO06GC-F5JR400N |
| JR4.0/6F | TCO06GC-F6JR400N |
| JR4.0/7F | TCO06GC-F7JR400N |
| MULTIPURPOSE 6F | TCO06GC-F6MP010N |
| TR4.0/6F | TCO06GC-F6TR400N |
Description
Optimize your challenging procedure
Heartrail™ II will improve your back-up force when using Ikari curves right and left during transradial interventions or through a 5Fr guiding catheter in 6Fr to increase backup support.
Characteristics
- Ikari curves right and left are developed to improve your back-up force during TRI via the right radial artery
- Tiger curves are designed to be used for both right and left coronary arteries
Feel the difference with the 5Fr guiding catheter in 6Fr
By inserting a 5 Fr (120 cm with flexible distal portion) into a 6 Fr guiding catheter:
- Provides the back-up support of a 7 Fr guiding catheter with a 6 Fr
- Minimizes puncture size
- Increases your back-up support for an excellent PTCA balloons crossing success rate1
General specifications
| Shaft inner layer material | Polytetrafluoroethylene – PTFE |
|---|---|
| Braiding | High strength flat wire mesh pattern |
| Inner diameter | 5 Fr – 0.059″ (1.50 mm) 6 Fr – 0.071″ (1.80 mm) 7 Fr – 0.081″ (2.06 mm) |
| Usable length | 100 cm 120 cm – Heartrail 5Fr straight, for 5Fr guiding catheter in 6Fr |
Item specifications
| Shape type | Shape code | 5 Fr | 6 Fr | 7 Fr |
|---|---|---|---|---|
| Amplatz Left | AL 1 | GC-K5AL010N | GC-K6AL010N | GC-K7AL010N |
| AL 2 | GC-K5AL020N | GC-K6AL020N | GC-K7AL020N | |
| AL 3 | GC-K5AL030N | GC-K6AL030N | GC-K7AL030N | |
| Amplatz Right | AR 1 | GC-K5AR010N | GC-K6AR010N | GC-K7AR010N |
| AR 2 | GC-K5AR020N | GC-K6AR020N | GC-K7AR020N | |
| Judkins Left | JL 3 | — | GC-K6JL300N | — |
| JL 3.5 | GC-K5JL350N | GC-K6JL350N | GC-K7JL350N | |
| JL 4 | GC-K5JL400N | GC-K6JL400N | GC-K7JL400N | |
| JL 4.5 | GC-K5JL450N | GC-K6JL450N | — | |
| JL 5 | GC-K5JL500N | GC-K6JL500N | GC-K7JL500N | |
| JL 6 | GC-K5JL600N | GC-K6JL600N | — | |
| Judkins Right | JR 3 | — | — | — |
| JR 3.5 | GC-K5JR350N | GC-K6JR300N | GC-K7JR350N | |
| JR 3.5 | — | GC-K6JR350N | — | |
| JR 4 | GC-K5JR400N | GC-K6JR400N | GC-K7JR400N | |
| JR 4.5 | GC-K5JR450N | — | — | |
| JR 5 | GC-K5JR500N | GC-K6JR500N | — | |
| JR 6 | — | GC-K6JR600N | — | |
| BackUp for left coronary |
BL 2.5 | — | — | — |
| BL 3 | GC-K5BL300N | GC-K6BL300N | GC-K7BL300N | |
| BL 3.5 | GC-K5BL350N | GC-K6BL350N | GC-K7BL350N | |
| BL 4 | GC-K5BL400N | GC-K6BL400N | GC-K7BL400N | |
| BL 4.5 | GC-K5BL450N | GC-K6BL450N | GC-K7BL450N | |
| BackUp for right coronary |
BR 3.5 | — | GC-K6BR350N | — |
| BR 4 | — | GC-K6BR400N | — | |
| IMA | IMA 1.0 | GC-K5IMA10N | GC-K6IMA10N | GC-K7IMA10N |
| Multipurpose | MPA-Large | GC-K5MP030N | GC-K6MP030N | GC-K7MP030N |
| Bypass |
BP-L | GC-K5BP010N | — | — |
| BP-R | GC-K5BP020N | GC-K6BP020N | — | |
| Ikari-Curve Left |
IL 3 | — | GC-K6IL300N | — |
| IL 3.5 | GC-K5IL350N | GC-K6IL350N | GC-K7IL350N | |
| IL 4 | GC-K5IL400N | GC-K6IL400N | GC-K7IL400N | |
| IL 4.5 | — | GC-K6IL450N | — | |
| Ikari-Curve Right |
IR 1 | — | GC-K6IR100N | — |
| IR 1.5 | GC-K5IR150N | GC-K6IR150N | GC-K7IR150N | |
| IR 2 | GC-K5IR200N | GC-K6IR200N | GC-K7IR200N | |
| Tiger | TIG 5 | GC-K5TR500N | — | — |
| — | — | — | — | GC-K7TR500N |
| — | — | — | GC-K6MP020N | — |
| — | — | — | GC-K6TR500N | — |
| — | — | — | GC-K6MP010N | — |
| — | — | — | GC-K6MP010N | — |
| 5Fr guiding catheter in 6Fr | Straight | GC-K5ST012NQ | — | — |
Heartrail® II – PTCA Guiding Catheter
Related products
-
EUPHORA SEMICOMPLIANT BALLOON CATHETER
Euphora™ semicompliant balloon
Balloon Dilatation Catheter
Euphora™ semicompliant balloon dilatation catheter complements your expertise and provides uncompromising performance in the moment you need it most.
OVERVIEW
Advancing Innovation through Partnership
To develop the Euphora™ low-profile semicompliant balloon dilatation catheter, we partnered with nearly 2,000 interventional cardiologists and cath lab professionals. The result is:
- Superior deliverability1
- Superb Kissing Balloons Technique (KBT) performance2
- The lowest crossing profile compared to leading competitors3
PRODUCT DETAILS
What Makes the Euphora Balloon an Excellent Choice?
ORDERING INFORMATION
BALLOON LENGTHS 6, 10, 12, AND 15 MM
1.50 EUP1506X EUP1510X EUP1512X EUP1515X 2.00 EUP2006X EUP2010X EUP2012X EUP2015X 2.25 EUP22506X EUP22510X EUP22512X EUP22515X 2.50 EUP2506X EUP2510X EUP2512X EUP2515X 2.75 EUP27506X EUP27510X EUP27512X EUP27515X 3.00 EUP3006X EUP3010X EUP3012X EUP3015X 3.25 EUP32506X EUP32510X EUP32512X EUP32515X 3.50 EUP3506X EUP3510X EUP3512X EUP3515X 3.75 EUP37506X EUP37510X EUP37512X EUP37515X 4.00 EUP4006X EUP4010X EUP4012X EUP4015X BALLOON LENGTHS 20, 25, AND 30 MM
1.50 EUP1520X — — 2.00 EUP2020X EUP2025X EUP2030X 2.25 EUP22520X EUP22525X — 2.50 EUP2520X EUP2525X EUP2530X 2.75 EUP27520X EUP27525X — 3.00 EUP3020X EUP3025X EUP3030X 3.25 EUP32520X EUP32525X — 3.50 EUP3520X EUP3525X EUP3530X 3.75 EUP37520X EUP37525X — 4.00 EUP4020X EUP4025X EUP4030X -
PTCA Balloon Ryurei
Description
Confidence in your hands
Ryurei™ has superior crossability1 thanks to the combination of low distal profiles and Terumo’s hydrophilic M coating. The short balloon bonding and reinforced shaft1 results in enhanced trackability1 while maintaining excellent pushability1.
These features can increase physician’s confidence in success even during complex procedures.
CharacteristicsExcellent Pusability
The combination of flexible distal part, tapered core wire on the midshaft and the thick proximal hypotube, results in smooth and balanced transition across the entire catheter for high transmission force, resulting in excellent pushability
Superior Crossability2
Thanks to the combination of distal low profiles and the Terumo hydrophilic M-Coat, Ryurei offers low friction in crossing tight lesions
Enhanced Trackability2
With an optimal tip design and improved balloon bonding, Ryurei provides enhanced trackability, even in tortuous anatomy and difficult to navigate vessels.
General specifications
Shaft diameter 1.9 Fr/0.64 mm Proximal • 2.5 Fr/0.84 mm Middle • 2.4 Fr/0.79 mm to 2.7 Fr/0.89 mm (tapered) Distal (1.00 to 1.50 mm) • 2.6 Fr/0.87 mm Distal (2.00 to 4.00 mm) Usable length 145 cm Balloon markers # 1(Ø1.0 to Ø 1.5 mm) • 2(Ø2.0 to Ø4.0 mm) Nominal pressure 6 atm / 608 kPa Rated burst pressure Ø1.0 – 3.0 mm=14 atm(1419kPa) • Ø3.25 – 4.0 mm = 12 atm(1216 kPa) Guidewire compatibility Maximum diameter: 0.014″ / 0.36 mm Lesion entry profile 0.41 mm (Ø1.00 to Ø1.50 mm) • 0.43 mm (Ø2.00 to Ø4.00 mm) Coating Hydrophilic Item specifications
Balloon length / Product code
Balloon Diameter 5 mm 10 mm 15 mm 20 mm 30 mm 40 mm 1.00 mm DC-RR1005HH 1.25 mm DC-RR1205HH DC-RR1210HH DC-RR1215HH DC-RR1220HH 1.50 mm DC-RR1505HH DC-RR1510HH DC-RR1515HH DC-RR1520HH 2.00 mm DC-RR2010HHW DC-RR2015HHW DC-RR2020HHW DC-RR2030HHW DC-RR2040HHW 2.25 mm DC-RR2210HHW DC-RR2215HHW DC-RR2220HHW 2.50 mm DC-RR2510HHW DC-RR2515HHW DC-RR2520HHW DC-RR2530HHW DC-RR2540HHW 2.75 mm DCRR2710HHW DC-RR2715HHW DC-RR2720HHW 3.00 mm DC-RR3010HHW DC-RR3015HHW DC-RR3020HHW DC-RR3030HHW DC-RR3040HHW 3.25 mm DC-RR3210HHW DC-RR3215HHW DC-RR3220HHW 3.50 mm DC-RR3510HHW DC-RR3515HHW DC-RR3520HHW DC-RR3530HHW DC-RR3540HHW 3.75 mm DC-RR3710HHW DC-RR3715HHW DC-RR3720HHW 4.00 mm DC-RR4010HHW DC-RR4015HHW DC-RR4020HHW References
1 Ryurei φ1.00-5mm compared to Tazuna 1.25-10 mm- Data on file ISCD-416-31-4
* For Ryurei Ø 1.00 mm – Ø 1.50 mm.
** For Ryurei Ø 1.00mm – 5mm 2 Compared to Tazuna PTCA dilatation catheter
-
RADIFOCUS INTRODUCER SHEATH
Radifocus™ Introducer II Standard Kit A
Introducer Sheath (FEMORAL)
Kit of vascular introducer with hemostatic valve with excellent blood tightness,
easy and non traumatic vessel insertion, patented valve design for a unique
features package.- Total Integrated Fit (TIF) tip tapering: tapering design at the tip of the sheath and dilator to facilitate smooth penetration
- Cross-cut hemostasis valve to avoid blood reflux and air aspiration
- Thin radiopaque sheath with anti-kinking sleeve for easy catheter handling
- Snap-on/click-off dilator lock prevents dilator back-out during insertion and allows one-hand unlocking
- Wide variety of kit variations providing all elements for quick vessel access: 4-11 Fr sheaths, 5-16 cm lengths, Surf lash needle
General specifications
Sheath length 10 cm Mini guidewire Plastic Straight and Angled
0.025″ (0.64 mm) for 4 Fr, straight mini guidewire type; 0.035″ (0.89 mm) for all others
45 cmEntry needle Plastic IV Catheter – 18G x 2 ½” (1.2 x 64 mm),
except for 4 Fr with 45 cm straight mini guidewire: 20G x 2″ (0.9 x 51 mm)
2.5 ml syringe is includedGuidewire compatibility 0.025″ (0.64 mm) for 4 Fr, straight mini guidewire type
0.035″ (0.89 mm) for all othersPackaging Tray A Kit content Sheath, dilator, plastic mini guidewire and plastic IV catheter (entry needle) Item specifications
Inner diameter Mini guidewire type 45 cm angled 45 cm straight 4 Fr RS+A40K10AQ RS+A40G10SQ 4 Fr RS*A40G10SQ RS*A40G05SQ 4 Fr RS*A40K10SQ RS*A40G07SQ 5 Fr RS*A50K10AQ RS*A50G07SQ 5 Fr RS+A50K10AQ RS+A50K10SQ 6 Fr RS+A60K10AQ RS+A60K10SQ 6 Fr RS*A60K10AQ RS*A60G07SQ 6 Fr — RS*A60G16SQZ 6 Fr — RS*A60K10SQ 6 Fr — RS+A50K10SQ 7 Fr — RS+A70K10SQ 7 Fr — RS*A70G07SQ 7 Fr — RS*A70K10SQ 8 Fr — RS+A80K10SQ 8 Fr — RS*A80K10SQ 9 Fr — RS*A90K10SQ 10 Fr — RS*A10K10SQ 11 Fr — RS*A11K10SQ A Kit includes: introducer sheath, dilator, mini Guide wire, entry venous catheter, 2.5 cc syringe
B Kit includes: introducer sheath, dilator, mini Guide wire
C Kit includes: introducer sheath, dilator
R Kit includes: introducer sheath, dilator, mini Guide wire, 18 G x 2 3/4” / 1.2 x 70 mm entry metallic needle
Other code numbers are available on special demand. For any further information, please contact your Terumo local representative
Units per box: 5 for kits in tray and 10 for kits in pouch. -
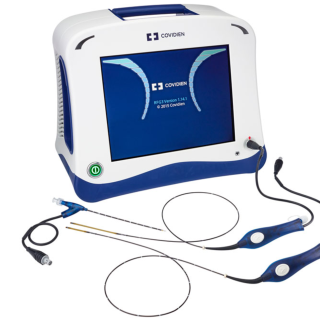
CLOSUREFAST
ClosureFast™
procedureMinimally invasive thermal
treatment for vein disease
and varicose veins.
The ClosureFast procedure uses radiofrequency energy or heat to close the diseased vein, which redirects blood flow to healthy veins, relieving symptoms.Before the procedure
You will have an ultrasound imaging exam of the leg being treated. This exam is important for assessing the diseased superficial vein and planning the procedure.
During the procedure
Your doctor will discuss the procedure with you. Here is a brief summary of what to expect:
- The ClosureFast procedure is performed on an outpatient basis. Your doctor will perform the ClosureFast procedure. Using an ultrasound, your doctor will position a catheter in the diseased vein through a small opening in the skin.
- The small catheter delivers heat to the vein wall, causing it to shrink and seal the vein. Once the diseased vein is closed, blood will reroute itself to other healthy veins.
After the procedure
- You will be taken to the recovery area to rest.
- Following the procedure, your doctor will apply a simple bandage over the insertion site and will give you compression stockings to aid in the healing process. Your doctor may encourage you to walk and to refrain from extended standing and strenuous activities for a period of time. The average patient typically resumes normal activities within a few days.1,2
- Your doctor will recommend follow-up care as needed.
-
Benefits of the ClosureFast procedure
- Relief of symptoms after two days, with a noticeable improvement in one to two week
- Minimally invasive outpatient procedure
- Less pain and bruising than laser treatment
- Faster recovery than laser treatment
- Proven results with positive patient experience
- Rapid recovery — on average, patients resume normal activities within a few days
- Lasting results — the only radiofrequency energy procedure with published long-term clinical data demonstrating safety and efficacy, with a 91.9% closure rate at five years.4
Adverse events can include nerve injury, hematoma, phlebitis, thrombosis, and/or pulmonary embolism.
Type of therapy
Thermal shrinkage of the vein wall with radiofrequency energy or heatInpatient or outpatient procedure
Outpatient procedureOne- versus two-leg treatment
Typically, each leg will be treated in separate appointmentsNeedle sticks required
One needle stick for vein access. Typically, 5 to 10 needle sticks for anesthesia.Typical post-treatment recovery
Healing of vein access site, anesthetic needle stick sites, and also healing of ablated vein sectionCompression hose
Required for approximately one weekProcedure success rate
91.9% after five years4
ClosureFast procedure FAQ
Is the ClosureFast procedure painful?
Most patients report feeling little, if any, pain during the ClosureFast procedure.3 Your doctor will give you a local or regional anesthetic to numb the treatment area.
When can I return to normal activity?
Patients treated with the ClosureFast procedure may resume normal activity more quickly than patients who undergo surgical vein stripping or laser ablation. With the ClosureFast procedure, the average patient typically resumes normal activity within a few days.2 For a few weeks following the treatment, your vein specialist may recommend a regular walking regimen and suggest you refrain from strenuous activities (heavy lifting, for example) or prolonged periods of standing.
When will my symptoms improve?
Most patients report relief of symptoms after two days, with a noticeable improvement in one to two weeks.
Is there any scarring, bruising, or swelling after the procedure?
Most patients report limited to no scarring, bruising, or swelling following the ClosureFast procedure.3
How is the ClosureFast procedure different from endovenous lasers?
Both Closure Fast and lasers use thermal technology to deliver heat into the diseased vein, but they each use a different method of delivery. Despite their similarity in using heat, a 2009 study showed that the ClosureFast procedure is associated with lower rates of pain, bruising, and complications and a faster improvement in patients’ quality of life when compared to 980 nm laser ablation.4
How is the Closure Fast procedure different from vein stripping?
During vein stripping, incisions are made in the groin and calf, and a tool is threaded through the diseased vein to pull the vein out of the leg. With the ClosureFast procedure, only one small incision is made at the insertion site and the vein is then treated and left in place. ClosureFast is a minimally invasive approach that reduces the likelihood of pain and bruising, associated with vein stripping surgery.1
Is the ClosureFast procedure covered by insurance?
Many patients have access to the ClosureFast procedure through their insurance coverage plan. Insurance companies detail access to the ClosureFast procedure and other radiofrequency ablation procedures in coverage policies for varicose veins or chronic venous insufficiency. As with all healthcare procedures, you may also have some cost associated with receiving treatment, such as a copay or coinsurance. Please reach out to your insurance company to discuss your specific plan coverage and potential costs prior to seeking treatment





Reviews
There are no reviews yet.