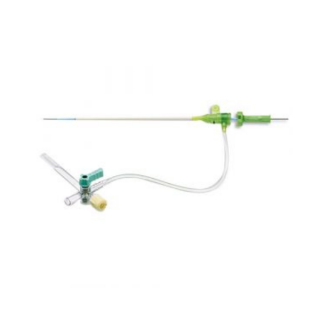
GUIDING CATHETER EKARI
Product Overview
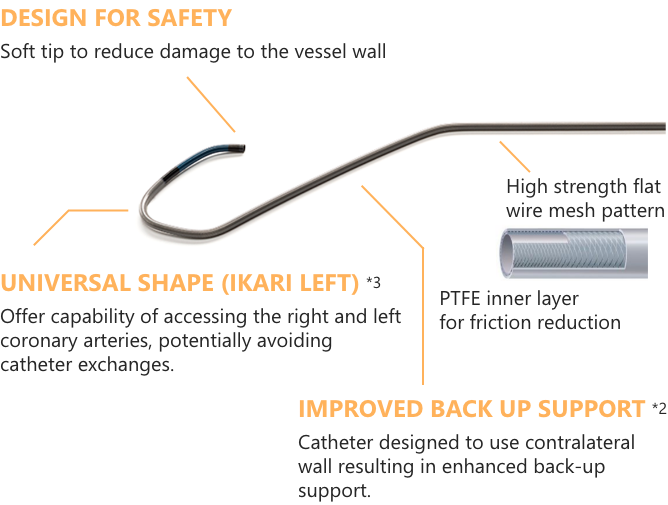
IMPROVED BACK UP SUPPORT*1
Catheter designed to use contralateral wall resulting in enhanced back-up support.
UNIVERSAL SHAPE (IKARI LEFT) *2
Offer capability of accessing the right and left coronary arteries, potentially avoiding catheter exchanges.
DESIGN FOR SAFETY
Soft tip to reduce damage to the vessel wall

*1: Ikari Y, Nagaoka M, Kim JY, Merino Y, Tanabe T. The physics of guiding catheters for the left coronary artery in transfemoral and trans-radial interventions. J Invasive Cardio. 2005 Dec; 17(12): 636-641.*2: Youssef AA, Hsieh YK, Cheng CI, We CJ. A single trans-radial guiding catheter for right and left coronary angiography and intervention. Euro intervention 2007; 3: 475-481.
Patients who have had cardiac catheterization or coronary angioplasty often are required to stay in bed with restricted movement for three to 24 hours afterward to prevent bleeding from the femoral artery catheter insertion site.
Features and Benefits
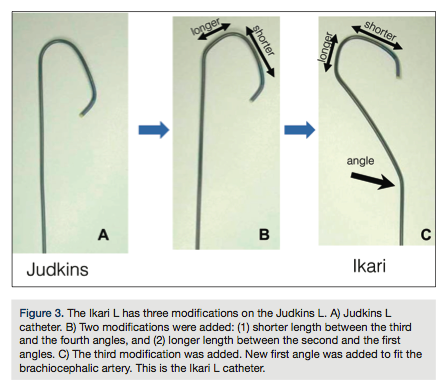
Improved back up support in comparison to Terumo standard femoral guiding catheter shapes*1

*1: Based on comparative studies made among Terumo Guiding Catheters*2: Ekari Y, Nagaoka M, Kim JY, Merino Y, Tanabe T. The physics of guiding catheters for the left coronary artery in transfemoral and trans-radial interventions. J Invasive Cardio. 2005 Dec; 17(12): 636-641.*3: Youssef AA, Hsieh YK, Cheng CI, We CJ. A single trans-radial guiding catheter for right and left coronary angiography and intervention. Euro intervention 2007; 3: 475-481.
Guide catheters are required for all coronary interventions to provide access to the coronary ostium and support equipment delivery. The ideal guide provides stability for device advancement through the coronary anatomy, while minimizing vessel trauma and allowing for vessel opacification.
The catheter is guided to the narrowed artery. Then, a smaller balloon catheter is inserted through the flexible catheter and inflated at the narrowed area to open it. Often, the doctor will also place a mesh coil called a stent at the narrowed part to help keep the artery open.
Related products
-
RADIFOCUS REDIAL SHEATH
Providing you the best range of Terumo Radifocus Radial Introducer Sheath and Cordis Femoral Introducer Sheath with effective & timely delivery.
The radial artery access sheath is used to gain arterial access and facilitate the insertion of catheters or other equipment for diagnostic and vascular interventions.
- The Cordis AVANTI®+ Introducer is the pioneer of catheter sheath introducer technology. Featuring a patented SLIX Valve, the AVANTI®+ introducer provides smooth transitions, monitoring capabilities and exceptional performance for your procedural success, as well as
- A hexacuspid design that provides a balance between catheter maneuverability and hemostasis.
- A rotating suture collar that facilitates procedural flexibility. It stays in place and allows patient movement.
- A kink-resistant cannula design integrating a soft, flexible inner layer with a stiffer outer layer designed to increase bendability and support, and decrease kinking.
- Atraumatic tip transitions for both the sheath and the vessel dilator are uniquely tapered and manicured. This results in smooth insertions and helps to minimize damage upon entry.
As part of the circulatory system, the radial artery supplies blood from the heart to the forearm.
Radial sheaths had the smallest OD (1 Fr smaller) when compared to similar sized conventional sheaths. For instance, the OD of 4 Fr radial sheath (1.610 ± 0.006 mm) is essentially the same as the OD of the conventional 3 Fr (1.644 ± 0.016 mm) and 3.3 Fr (1.635 ± 0.005 mm) sheaths.
-
NC Sprinter PTCA BALLOON
NC Sprinter RX Noncompliant
NC Stormer over-the-wire noncompliant balloon dilatation catheter offers controlled compliance, high pressures, and comprehensive lengths.
Balloon Dilatation Catheter
OVERVIEW
The NC Sprinter™ RX noncompliant coronary balloon combines:
- Sprinter crossing technology
- High-pressure capability and controlled expansion1
ORDERING INFORMATION
BALLOON LENGTHS 6, 9, 12 MM
Balloon Diameter (mm)
Balloon Length (mm) 6 9 12 2.00 NCSP2006X NCSP2009X NCSP2012X 2.25 NCSP22506X — NCSP22512X 2.50 NCSP2506X NCSP2509X NCSP2512X 2.75 NCSP27506X NCSP27509X NCSP27512X 3.00 NCSP3006X NCSP3009X NCSP3012X 3.25 NCSP32506X NCSP32509X NCSP32512X 3.50 NCSP3506X NCSP3509X NCSP3512X 3.75 NCSP37506X NCSP37509X NCSP37512X 4.00 NCSP4006X NCSP4009X NCSP4012X 4.50 — — — 5.00 — — — BALLOON LENGTHS 15, 21, 27 MM
Balloon Diameter (mm)
Balloon Length (mm) 15 21 27 2.00 NCSP2015X NCSP2021X — 2.25 — NCSP22521X — 2.50 NCSP2515X NCSP2521X NCSP2527X 2.75 NCSP27515X NCSP27521X — 3.00 NCSP3015X NCSP3021X NCSP3027X 3.25 NCSP32515X NCSP32521X — 3.50 NCSP3515X NCSP3521X NCSP3527X 3.75 NCSP37515X NCSP37521X — 4.00 NCSP4015X NCSP4021X NCSP4027X 4.50 NCSP4515X NCSP4521X — 5.00 NCSP5015X — — -
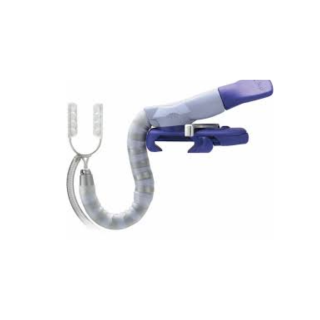
OCTOPUS TISSUE STABILIZER
Octopus
Tissue Stabilizers
OCTOPUS TISSUE STABILIZERS
Our newest generation tissue stabilizer features automatic pod spread for effective visualization.
SUMMARY
OCTOPUS EVOLUTION AS – THE NEXT GENERATION TISSUE STABILIZER
The newest generation tissue stabiliser from Medtronic demonstrates an enduring commitment to the cardiac surgeon. With all the same features of the Octopus™ Evolution – enhanced stability, flexibility, and a lower profile for unlimited positioning options – the Octopus Evolution AS offers an automatic pod spread feature that enhances visibility at the anastomotic site.
- Automatic pod spread for effective visualisation of the anastomotic site
- Very secure arm for maximum stabilization
- Greater flexibility enables unlimited positioning options
- Simple, secure, one-handed attachment of clamp to the retractor
OCTOPUS EVOLUTION – LOW PROFILE WITH FLEXIBILITY AND REACH
The Octopus Evolution stabilizer provides enhanced stability, flexibility, and a lower profile for unlimited positioning options.
Enhanced stability and flexibility features:
- Improved visualisation with:
- Lower profile headlink
- Surgeon controlled pod-spread
- Allowance for multiple device positions including pods-up, pods-down and pods-to-the-side applications
- Increased flexibility and range of motion
- Longer, effective reach of the articulating arm provides easier access to all vessels
- Simple, secure, one-handed engagement of clamp and turret assembly
THE OCTOPUS 4.3 STABILISER FEATURES SUPERIOR ARM STRENGTH AND UNMATCHED STABILITY IN THE CLAMP/TURRET MECHANISM.
- Dual vacuum tubes offer superior tissue capture
- Internal cable enables arm twisting for maximum access and flexibility
- Reinforced arm links offer better stabilisation
- Whale tail tightens easily
- Swivel turret offers access while maximising visualisation
- Head lock design allows “toes up” position, pod spread and bend
- Rigid clamp designed to eliminate rocking
OCTOPUS 4 – EXCELLENT STABILITY AND FLEXIBILITY
The Octopus 4 tissue stabiliser includes innovative features, such as clear, malleable pods combined with a smaller diameter, more flexible arm, and an ultra-low headlink profile.
- Pod spreading optimises sewing space and stability of anastomotic site
- Reduced headlink profile improves visibility of surgical site
- Rigid, multi-material arm reduces movement at the anastomotic site
- Smaller diameter, more flexible arm offers greater flexibility in positioning
- Smaller, color-coded whale tail provides minimal obstruction
- Single vacuum tubing line minimises obstruction
- Innovative turret design increases effective arm reach, dramatically increasing positioning options with 360-degree movement
-
PROGREAT® MICROCATHETHER
PRODUCT OVERVIEW
PROGREAT® Microcatheters are designed to allow navigation through tortuous peripheral vessels for optimal access and delivery of therapeutic embolization.
Terumo Progreat 2.7Fr. Microcatheter is Designed to provide optimal trackability and support even in the most tortuous anatomy.
DESIGNED TO CONFIDENTLY DELIVER EMBOLOTHERAPY
- Tungsten coil reinforcement for kink resistance2
- PTFE inner layer is designed for exceptional guidewire tracking, with virtually frictionless delivery of coils and other embolics2
- DMSO Compatible3
- Radiopaque 0.7 mm platinum/iridium markers allow for rapid and precise positioning4
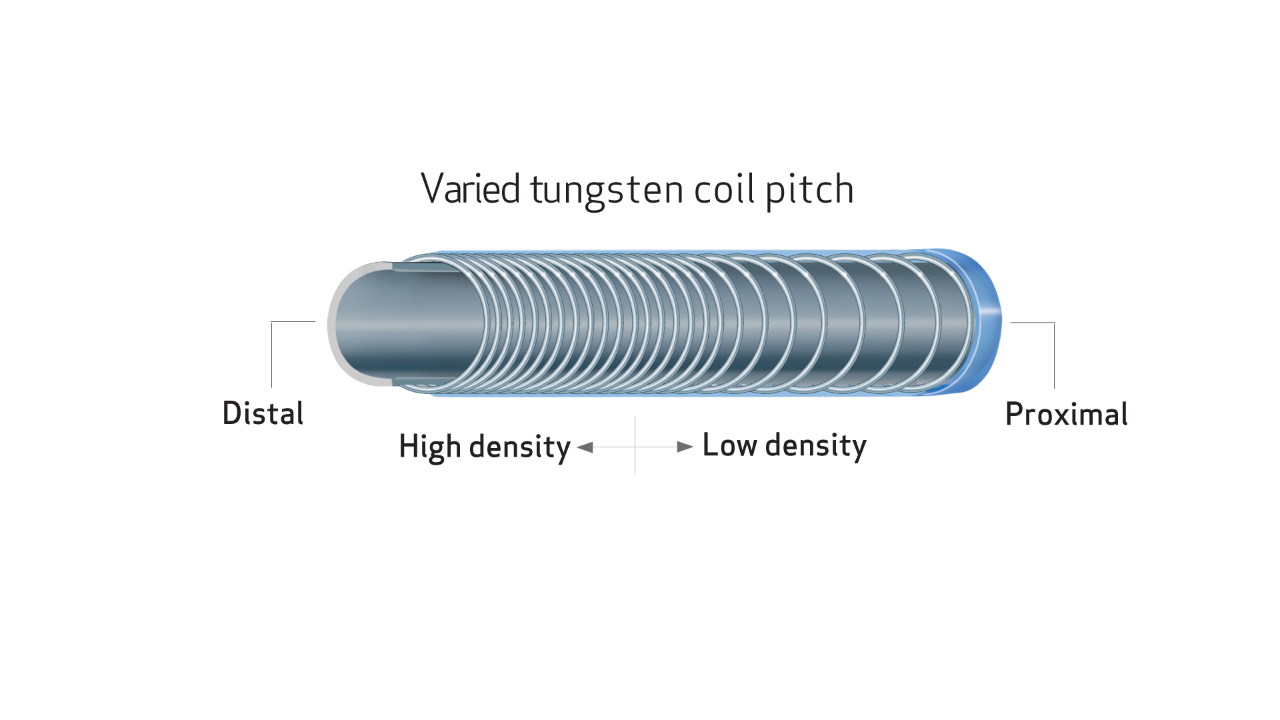
DESIGNED TO ENHANCE ACCESS TO SMALL PERIPHERAL VESSELS
- TERUMO Glide TechnologyTM hydrophilic coating enhances navigation through tortuous anatomy1,2
- Unique varied tungsten coil pitch construction provides distal flexibility and proximal pushability, enhancing vessel selectivity and catheter placement.
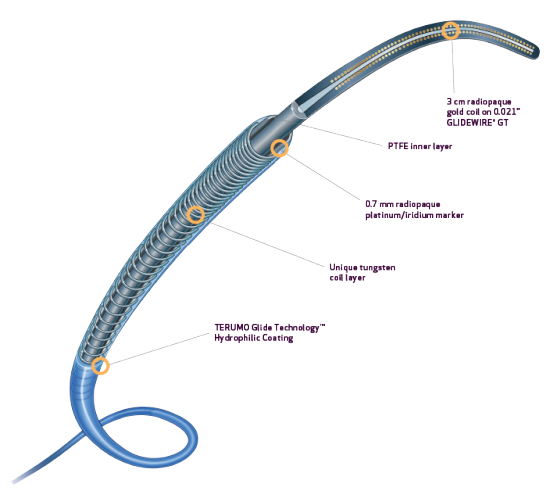
PROGREAT® COAXIAL MICROCATHETER SYSTEM
- Preloaded with uniquely designed 0.021” GLIDEWIRE® GT for the 2.8Fr system and 0.018″ GLIDEWIRE® GT for the 2.4Fr system1
- Available in 2.8Fr and 2.4Fr
- Enables simultaneous preparation of microcatheter and guidewire, which may save time and increase procedural efficiency1
PROGREAT 2.4 COAXIAL PROVEN TO REACH DISTAL VESSELS WITH EASE⁴
- Provides greater vessel selectivity and demonstrated superiority in traversing tortuous anatomy
- Provides best-in-class kink resistance, demonstrated in both the distal and proximal shaft.

PROGREAT® Microcatheter Product Codes
Includes hemostatic valve and shaping mandrel
Fr Size Product Code Length (cm) Tip Shape Max Pressure (psi) RO Markers Hydrophilic Coating Length (cm) 2.4 MC*PB2411Y 110 Straight 750 1 50 MC*PB2413Y 130 Straight 1 70 MC*PB2413ZRA 130 70 Deg. Angle 1 70 MC*PB2413ZRC 130 J Curve 1 70 MC*PV2415Y 150 Straight 2 90 MC*PV2415ZRA 150 70 Deg. Angle 2 90 MC*PV2415ZRC 150 J Curve 2 90 2.7 MC*PC2711Y 110 Straight 750 – 50 MC*PC27137 130 Straight – 70 2.8 MC*PB2811Y 110 Straight 900 1 50 MC*PB2813Y 130 Straight 1 70 MC*PV2815Y 150 Straight 2 90 PROGREAT® Coaxial Microcatheter System Codes
Includes unique GLIDEWIRE® GT Guidewire, wire stopper, guidewire introducer, hemostatic valve, 2.5 mL syringe, and shaping mandrel
Fr Size Product
CodeLength
(cm)Max Pressure
(PSI)RO Markers Hydrophilic
Coating Length
(cm)Glidewire® GT
Length
(cm)Glidewire® GT Size
(in)2.4 Fr MC*PE24111YB 110 750 1 50 120 0.018 2.4 Fr MC*PE24131YB 130 750 1 70 140 0.018 2.4 Fr MC*PE24151YV 150 750 2 90 160 0.018 2.7 Fr MC*PE27111Y 110 750 – 50 120 0.021 2.7 Fr MC*PE27131Y 130 750 – 70 140 0.021 2.8 Fr MC*PE28111YB 110 900 1 50 120 0.021 2.8 Fr MC*PE28131YB 130 900 1 70 140 0.021 2.8 Fr MC*PE28151YV 150 900 2 90 160 0.021 PRODUCT SPECIFICATIONS
Catheter OD Length
(cm)Inner Diameter
(in/mm)Max GW
(in)Embolic
CompatibilityDead Space Volume
(mL)Actual Flow Rate* (mL/sec) @ 750 psi Actual Flow Rate* (mL/sec) @ 900 psi 2.4/2.9Fr
(0.80/0.97mm)110 0.022″/0.57 0.018″ 0.018″ Coils/HydroPearl®
600 ± 75 μm0.48 2.3 – 2.4/2.9Fr
(0.80/0.97mm)130 0.022″/0.57 0.018″ 0.018″ Coils/HydroPearl®
600 ± 75 μm0.53 2 – 2.4/2.9Fr
(0.80/0.97mm)150 0.022″/0.57 0.018″ 0.018″ Coils/HydroPearl®
600 ± 75 μm0.58 1.8 – 2.7/2.9Fr
(0.90/0.97mm)110 0.025″/0.065 0.021″ 0.018″ Coils/HydroPearl®
800 ± 75 μm0.57 3.5 – 2.7/2.9Fr
(0.90/0.97mm)130 0.025″/0.065 0.021″ 0.018″ Coils/HydroPearl®
800 ± 75 μm0.64 3.2 – 2.8/3.0Fr
(0.93/1.00mm)110 0.027″/0.70 0.021″ 0.018″ Coils/HydroPearl®
800 ± 75 μm0.61 4.1 4.7 2.8/3.0Fr
(0.93/1.00mm)130 0.027″/0.70 0.021″ 0.018″ Coils/HydroPearl®
800 ± 75 μm0.68 3.7 4.3 2.8/3.0Fr
(0.93/1.00mm)150 0.027″/0.70 0.021″ 0.018″ Coils/HydroPearl®
800 ± 75 μm0.75 3.1 3.9 REFERENCES
RX ONLY. Refer to the product labels and package insert for complete warnings, precautions, potential complications, and instructions for use.
- Catheter [Instructions for Use]. Somerset, NJ: Terumo Medical Corporation; 2016
- PROGREAT Catheter 510(k); 2003
- PROGREAT DMSO Compatibility Statement Letter
- Data on file.





Reviews
There are no reviews yet.