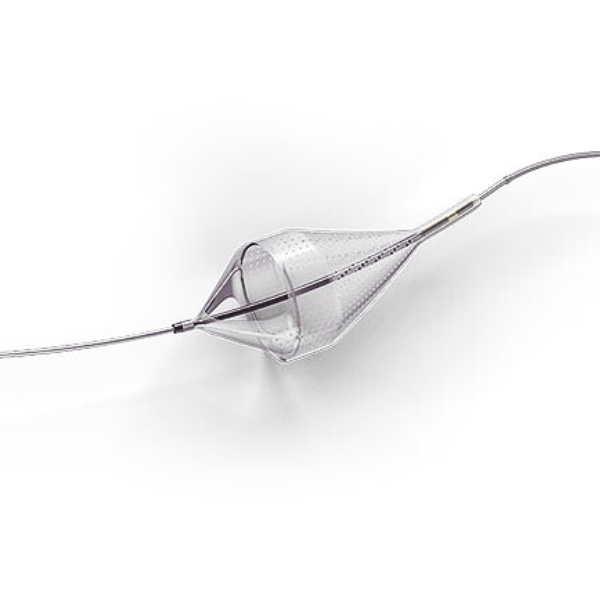
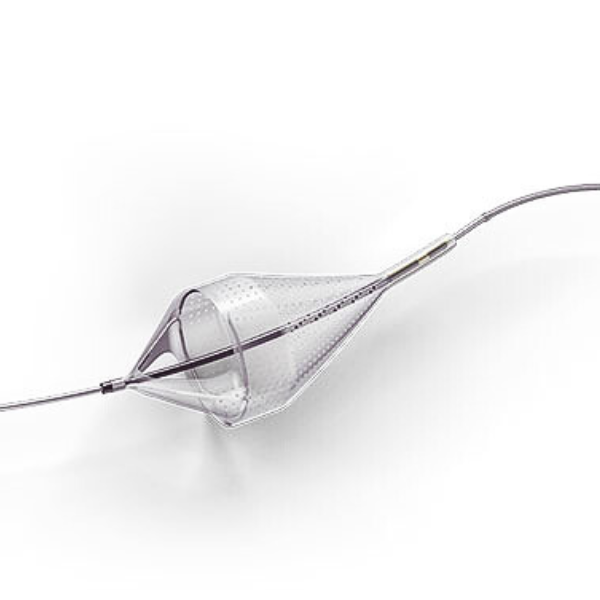
Emboshield NAV6 Embolic Protection System
Emboshield NAV6 Embolic
Indicated for Carotids and Lower Extremities
The Emboshield NAV6™ Embolic Protection System, which includes BareWire™ Filter Delivery Wires, allows the guide wire to rotate and advance freely, independent of the Emboshield NAV6™ filter.*
The Emboshield NAV6™ EPS is indicated for use as a guide wire and embolic protection system to contain and remove embolic material (thrombus / debris) while performing angioplasty and stenting procedures in carotid arteries and while performing atherectomy, during standalone procedures or together with PTA and/or stenting, in lower extremity arteries. The diameter of the artery at the site of the Filtration Element placement should be between 2.5 and 7.0 mm.
EMBOLIC PROTECTION DEVICE FOR BOTH LOWER EXTREMITY AND CAROTID ARTERIES
Captures Effectively to Minimize Downstream Complications1-3
- Centered wire design prevents bias against the vessel wall for effective debris capture.*
- Circumferential nitinol frame maintains optimal wall apposition, even on a bend.*
- Platinum-tungsten frame coils provide excellent visibility.*
- Two sizes allow for easy selection and minimal lab inventory.
Limits Filter Movement and Maintains Wire Access Through Innovative BareWire™ Design*
- The unique BareWire™ technology allows the wire to rotate and advance freely, independently of the filter.
- The filter is designed to stay in place during device delivery.
- Continued wire access, after filter is fully retracted, allows for easy delivery of additional therapy.
Navigates Skillfully Through Carotid and Lower Extremity Vasculature*
- Various types of BareWires™—distal access, workhorse, and support—are designed for various carotid anatomies.
- They promote navigational success through torturous anatomy and challenging arches.
Is Compatible with Various Atherectomy Options4
- Emboshield NAV6™ EPS and BareWire™ are compatible with a variety of atherectomy types.
- BareWire™ is available in 190 cm and 315 cm lengths.
In carotid procedures, the Emboshield NAV6™ EPS is used during stenting with the RX Acculink™ Carotid Stent System and the Xact™ Carotid Stent System.
* Data on file at Abbott.
References
- Bioangiu et al. Comparative analysis of retrieved particulate debris after superficial femoral atherectomy using three different atherectomy methods. EuroIntervention, May 2012.
- Bioangiu et al. Analysis of Retrieved Particulate Debris after Superficial Femoral Artery (SFA) Atherectomy Using the Pathway Jetstream G3 device. CCI, May 2011, 77(2) p. S57.
- Mendes et al. Clinical significance of embolic events in patients undergoing endovascular femoropopliteal interventions with or without embolic protection devices. JVS, February 2014, 59(2), 359-367.
- Philips Turbo-Elite, Medtronic TurboHawk PPES, and Boston Scientific Jetstream XC and SC. Test(s) performed by and data on file at Abbott.
IMPORTANT SAFETY INFORMATION
 Emboshield NAV6 Embolic Protection System
Emboshield NAV6 Embolic Protection System
Indications
The Emboshield NAV6 Embolic Protection System is indicated for use as a guide wire and embolic protection system to contain and remove embolic material (thrombus / debris) while performing angioplasty and stenting procedures in carotid arteries and while performing atherectomy, during standalone procedures or together with PTA and/or stenting, in lower extremity arteries. The diameter of the artery at the site of the Filtration Element placement should be between 2.5 and 7.0 mm.
Contraindications
The Emboshield NAV Embolic Protection System is contraindicated for use in
- Patients in whom anticoagulant and / or antiplatelet therapy is contraindicated.
- Patients with severe vascular tortuosity or anatomy that would preclude the safe introduction of the Guiding Catheter / Introducer Sheath, Embolic Protection System.
- Patients with a known allergy or hypersensitivity to device materials (Nitinol, Nickel, Titanium) or contrast medium, who cannot be adequately premedicated.
- Patients with uncorrected bleeding disorders.
- Lesions in the ostium of the common carotid artery.
- Inability to cross the lesion with the BareWire™ Filter Delivery Wire.
- Diffusely diseased vessels where there is no disease-free section in which to deploy the Filtration Element
- Insufficient straight section of vessel distal to the lesion to permit Filtration Element deployment.
Emboshield NAV6 Embolic Protection System
Related products
-
RESOLUTE INTEGRITY STENT
Resolute Onyx DES
for coronary artery disease
Resolute Onyx™ is a drug-eluting stent (DES) that’s different by design, optimised for complex PCI, and proven safe and effective in real-world, high bleeding risk patients on 1-month dual antiplatelet therapy (DAPT).1
Resolute Onyx DES is different by design to address your DES needs and a wide range of patient anatomies. It features:
- Best-in-class deliver ability
- Enhanced visibility with thinner struts
- Smooth side branch access
- Fast healing
- The broadest diameter range
Best-in-class deliverability
Single-wire design
Resolute Onyx DES is made from a single wire, which gives it a fluid range of motion and provides the flexibility needed for best-in-class deliverability.2

Sinusoid-formed wire

Helical wrap

Laser-fused
Competitive comparison
Resolute Onyx DES is more deliverable than laser-cut SYNERGY™* XD and XIENCE Skypoint™*, which are stiffer.
Deliverability comparison — 3.0 mm DES2
(Lower is better)

Enhanced visibility with thinner struts
Platinum-iridium core within Resolute Onyx DES increases visibility for accurate stent placement without compromising strut thickness.2Average visibility comparison2
(Higher is better)

- Cobalt alloy shell
- Platinum iridium core

Resolute Onyx cobalt alloy shell and platinum-iridum core
Smooth side branch access
Resolute Onyx DES has round struts to create a smooth passage when accessing the side branch and lower the propensity to catch during bifurcation procedures.2
Strut comparison
Rounded strut cross-section
- Resolute Onyx DES

Square strut cross-section
- SYNERGY™* DES
- XIENCE™* DES

Broadest diameter range
- Complex PCI requires a broad DES size matrix to match a wide range of patient anatomies.
- Resolute Onyx is the only DES with diameters ranging from 2.0 mm to 5.0 mm to treat a broad range of coronary vessel sizes.
Diameter (mm) Stent Length (mm) Maximum Expansion
Capabilities (MSID†) (mm)2.00 8 12 15 18 22 26 30 — — 3.50‡ 2.25 8 12 15 18 22 26 30 34 38 3.50‡ 2.50 8 12 15 18 22 26 30 34 38 3.50‡ 2.75 8 12 15 18 22 26 30 34 38 4.00‡ 3.00 8 12 15 18 22 26 30 34 38 4.00‡ 3.50 8 12 15 18 22 26 30 34 38 5.00‡ 4.00 8 12 15 18 22 26 30 34 38 5.00‡ 4.50 — 12 15 18 22 26 30 — — 6.00‡ 5.00 — 12 15 18 22 26 30 — — 6.00‡ PRODUCT DETAILS
2.00 RONYX20008X RONYX20012X RONYX20015X RONYX20018X RONYX20022X RONYX20026X RONYX20030X N/A N/A 2.25 RONYX22508X RONYX22512X RONYX22515X RONYX22518X RONYX22522X RONYX22526X RONYX22530X RONYX22534X RONYX22538X 2.50 RONYX25008X RONYX25012X RONYX25015X RONYX25018X RONYX25022X RONYX25026X RONYX25030X RONYX25034X RONYX25038X 2.75 RONYX27508X RONYX27512X RONYX27515X RONYX27518X RONYX27522X RONYX27526X RONYX27530X RONYX27534X RONYX27538X 3.00 RONYX30008X RONYX30012X RONYX30015X RONYX30018X RONYX30022X RONYX30026X RONYX30030X RONYX30034X RONYX30038X 3.50 RONYX35008X RONYX35012X RONYX35015X RONYX35015X RONYX35022X RONYX35026X RONYX35030X RONYX35034X RONYX35038X 4.00 RONYX40008X RONYX40012X RONYX40015X RONYX40018X RONYX40022X RONYX40026X RONYX40030X RONYX40034X RONYX40038X 4.50 N/A RONYX45012X RONYX45015X RONYX45018X RONYX45022X RONYX45026X RONYX45030X N/A N/A 5.00 N/A RONYX50012X RONYX50015X RONYX50018X RONYX50022X RONYX50026X RONYX5 -
ACT CARTRIDGE (HR-ACT)
Medtronic 402-03 ACT PLUS Disposable Test Cartridges 2-Channel Test Cartridges, High Range Activated Clotting Time (HR ACT) – Fresh whole blood test for CV surgery, PTCA, or vascular surgery, Box of 50
SKU:402-03Weight:3.00 LBSAvailability:2-3 Business daysShipping:Calculated at checkoutQuantity:Product Description
Medtronic 402-03 ACT PLUS Disposable Test Cartridges 2-Channel Test Cartridges, High Range Activated Clotting Time (HR ACT) – Fresh whole blood test for CV surgery, PTCA, or vascular surgery, Box of 50
ACT PLUS Disposable Test Cartridges 2-Channel Test Cartridges
402-01 Low Range Activated Clotting Time (LR ACT) – Fresh whole blood test for therapeutic heparin monitoring, post-angioplasty or dialysis 50 per box 402-02 Recalcified Activated Clotting Time (RACT) – For use with citrated whole blood samples at concentrations of 0.0 – 1.5 u/mL 50 per box 402-03 High Range Activated Clotting Time (HR ACT) – Fresh whole blood test for CV surgery, PTCA, or vascular surgery 50 per box 402-07 Heparinase Test Cartridge (HTC) – Fresh whole blood test used to identify the presence of heparin in the patient’s blood sample 20 per box -
Coronary Diagnostic Catheter (OPTITORQUE)
PRODUCT OVERVIEW
A complete line of coronary diagnostic catheters designed for superior torque control and precise placement.
RADIAL-SPECIFIC UNIVERSAL SHAPES
- Radial Specific curves designed to enable support and alignment during catheterization1
- Excellent back-up support for angiography of both RCA and LCA with a single catheter1,a
- Universal design eliminates the need for catheter exchanges and potentially shortens procedural and fluoroscopic time
1. DOF Reference
a. when compared to leading competitors
2. Ikaria article.OPTITORQUE Classic Shapes
Shape Category Product Code
5 Fr / 1.70 mmProduct Code
6 Fr / 2.00 mmShape Name (cm) Length Holes Side Tip Shape Pigtail 40-5030 40-6030 Straight Pigtail 110 6 40-5031 40-6031 Angled Pigtail 155° 110 6 Judkins Left 40-5040 40-6040 JL 3.5 (Judkins Left 3.5 cm) 100 40-5041 40-6041 JL 4.0 (Judkins Left 4.0 cm) 100 40-5042 40-6042 JL 5.0 (Judkins Left 5.0 cm) 100 Judkins Right 40-5043 40-6043 JR 3.5 (Judkins Right 3.5 cm) 100 40-5044 40-6044 JR 4.0 (Judkins Right 4.0 cm) 100 40-5045 JR 5.0 (Judkins Right 5.0 cm) 100 Bypass 40-5050 40-6050 Internal Mammary 100 Amplatz 40-5060 AL 1 (Amplatz Left Small Type) 100 40-5061 40-6061 AL 2 (Amplatz Left Middle Type) 100 UNIQUE DESIGN
- Designed for coaxial alignment to the ostium, allowing the catheter to sit at the appropriate angle for optimal engagement
- Large lumen allows high flow of contrast media and allows downsizing of French size
PROVEN PERFORMANCE
- Superior torqueability for precise placement1,a
- Excellent trackability in tortuous anatomy while navigating the radial artery1,a
- Superior flow rates for diagnostic imaging1,a
1.DOF
a. When compared to leading competitors -
NC Sprinter PTCA BALLOON
NC Sprinter RX Noncompliant
NC Stormer over-the-wire noncompliant balloon dilatation catheter offers controlled compliance, high pressures, and comprehensive lengths.
Balloon Dilatation Catheter
OVERVIEW
The NC Sprinter™ RX noncompliant coronary balloon combines:
- Sprinter crossing technology
- High-pressure capability and controlled expansion1
ORDERING INFORMATION
BALLOON LENGTHS 6, 9, 12 MM
Balloon Diameter (mm)
Balloon Length (mm) 6 9 12 2.00 NCSP2006X NCSP2009X NCSP2012X 2.25 NCSP22506X — NCSP22512X 2.50 NCSP2506X NCSP2509X NCSP2512X 2.75 NCSP27506X NCSP27509X NCSP27512X 3.00 NCSP3006X NCSP3009X NCSP3012X 3.25 NCSP32506X NCSP32509X NCSP32512X 3.50 NCSP3506X NCSP3509X NCSP3512X 3.75 NCSP37506X NCSP37509X NCSP37512X 4.00 NCSP4006X NCSP4009X NCSP4012X 4.50 — — — 5.00 — — — BALLOON LENGTHS 15, 21, 27 MM
Balloon Diameter (mm)
Balloon Length (mm) 15 21 27 2.00 NCSP2015X NCSP2021X — 2.25 — NCSP22521X — 2.50 NCSP2515X NCSP2521X NCSP2527X 2.75 NCSP27515X NCSP27521X — 3.00 NCSP3015X NCSP3021X NCSP3027X 3.25 NCSP32515X NCSP32521X — 3.50 NCSP3515X NCSP3521X NCSP3527X 3.75 NCSP37515X NCSP37521X — 4.00 NCSP4015X NCSP4021X NCSP4027X 4.50 NCSP4515X NCSP4521X — 5.00 NCSP5015X — —















Reviews
There are no reviews yet.