Emboshield NAV6 Embolic Protection System
Emboshield NAV6 Embolic
Indicated for Carotids and Lower Extremities
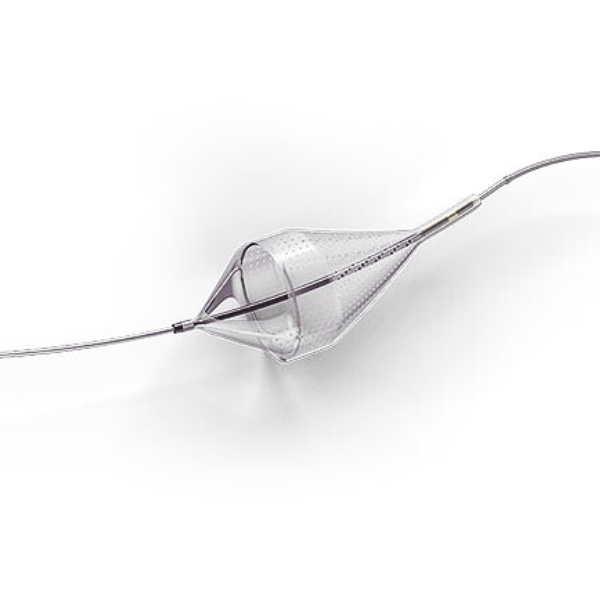
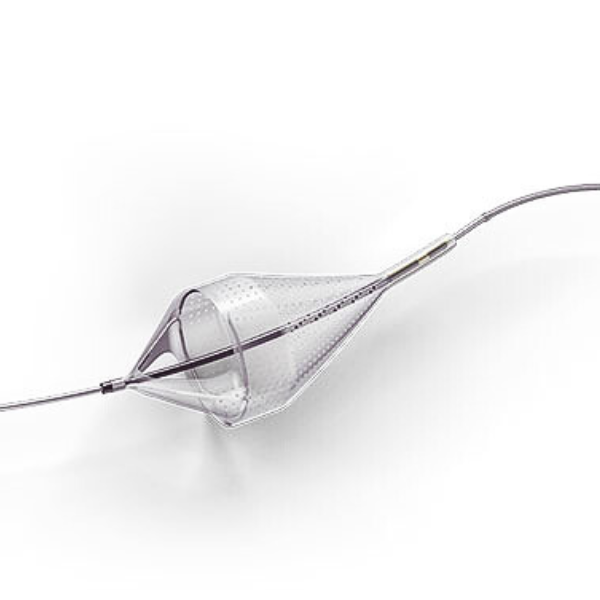
The Emboshield NAV6™ Embolic Protection System, which includes BareWire™ Filter Delivery Wires, allows the guide wire to rotate and advance freely, independent of the Emboshield NAV6™ filter.*
The Emboshield NAV6™ EPS is indicated for use as a guide wire and embolic protection system to contain and remove embolic material (thrombus / debris) while performing angioplasty and stenting procedures in carotid arteries and while performing atherectomy, during standalone procedures or together with PTA and/or stenting, in lower extremity arteries. The diameter of the artery at the site of the Filtration Element placement should be between 2.5 and 7.0 mm.
EMBOLIC PROTECTION DEVICE FOR BOTH LOWER EXTREMITY AND CAROTID ARTERIES
Captures Effectively to Minimize Downstream Complications1-3
- Centered wire design prevents bias against the vessel wall for effective debris capture.*
- Circumferential nitinol frame maintains optimal wall apposition, even on a bend.*
- Platinum-tungsten frame coils provide excellent visibility.*
- Two sizes allow for easy selection and minimal lab inventory.
Limits Filter Movement and Maintains Wire Access Through Innovative BareWire™ Design*
- The unique BareWire™ technology allows the wire to rotate and advance freely, independently of the filter.
- The filter is designed to stay in place during device delivery.
- Continued wire access, after filter is fully retracted, allows for easy delivery of additional therapy.
Navigates Skillfully Through Carotid and Lower Extremity Vasculature*
- Various types of BareWires™—distal access, workhorse, and support—are designed for various carotid anatomies.
- They promote navigational success through torturous anatomy and challenging arches.
Is Compatible with Various Atherectomy Options4
- Emboshield NAV6™ EPS and BareWire™ are compatible with a variety of atherectomy types.
- BareWire™ is available in 190 cm and 315 cm lengths.
In carotid procedures, the Emboshield NAV6™ EPS is used during stenting with the RX Acculink™ Carotid Stent System and the Xact™ Carotid Stent System.
* Data on file at Abbott.
References
- Bioangiu et al. Comparative analysis of retrieved particulate debris after superficial femoral atherectomy using three different atherectomy methods. EuroIntervention, May 2012.
- Bioangiu et al. Analysis of Retrieved Particulate Debris after Superficial Femoral Artery (SFA) Atherectomy Using the Pathway Jetstream G3 device. CCI, May 2011, 77(2) p. S57.
- Mendes et al. Clinical significance of embolic events in patients undergoing endovascular femoropopliteal interventions with or without embolic protection devices. JVS, February 2014, 59(2), 359-367.
- Philips Turbo-Elite, Medtronic TurboHawk PPES, and Boston Scientific Jetstream XC and SC. Test(s) performed by and data on file at Abbott.
IMPORTANT SAFETY INFORMATION
 Emboshield NAV6 Embolic Protection System
Emboshield NAV6 Embolic Protection System
Indications
The Emboshield NAV6 Embolic Protection System is indicated for use as a guide wire and embolic protection system to contain and remove embolic material (thrombus / debris) while performing angioplasty and stenting procedures in carotid arteries and while performing atherectomy, during standalone procedures or together with PTA and/or stenting, in lower extremity arteries. The diameter of the artery at the site of the Filtration Element placement should be between 2.5 and 7.0 mm.
Contraindications
The Emboshield NAV Embolic Protection System is contraindicated for use in
- Patients in whom anticoagulant and / or antiplatelet therapy is contraindicated.
- Patients with severe vascular tortuosity or anatomy that would preclude the safe introduction of the Guiding Catheter / Introducer Sheath, Embolic Protection System.
- Patients with a known allergy or hypersensitivity to device materials (Nitinol, Nickel, Titanium) or contrast medium, who cannot be adequately premedicated.
- Patients with uncorrected bleeding disorders.
- Lesions in the ostium of the common carotid artery.
- Inability to cross the lesion with the BareWire™ Filter Delivery Wire.
- Diffusely diseased vessels where there is no disease-free section in which to deploy the Filtration Element
- Insufficient straight section of vessel distal to the lesion to permit Filtration Element deployment.
Emboshield NAV6 Embolic Protection System
Related products
-
EUPHORA SEMICOMPLIANT BALLOON CATHETER
Euphora™ semicompliant balloon
Balloon Dilatation Catheter
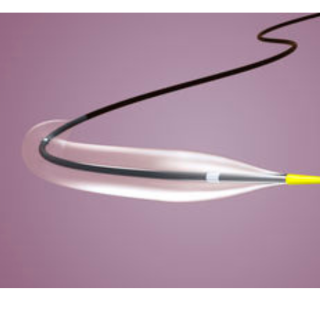
Euphora™ semicompliant balloon dilatation catheter complements your expertise and provides uncompromising performance in the moment you need it most.
OVERVIEW
Advancing Innovation through Partnership
To develop the Euphora™ low-profile semicompliant balloon dilatation catheter, we partnered with nearly 2,000 interventional cardiologists and cath lab professionals. The result is:
- Superior deliverability1
- Superb Kissing Balloons Technique (KBT) performance2
- The lowest crossing profile compared to leading competitors3
PRODUCT DETAILS
What Makes the Euphora Balloon an Excellent Choice?
ORDERING INFORMATION
BALLOON LENGTHS 6, 10, 12, AND 15 MM
1.50 EUP1506X EUP1510X EUP1512X EUP1515X 2.00 EUP2006X EUP2010X EUP2012X EUP2015X 2.25 EUP22506X EUP22510X EUP22512X EUP22515X 2.50 EUP2506X EUP2510X EUP2512X EUP2515X 2.75 EUP27506X EUP27510X EUP27512X EUP27515X 3.00 EUP3006X EUP3010X EUP3012X EUP3015X 3.25 EUP32506X EUP32510X EUP32512X EUP32515X 3.50 EUP3506X EUP3510X EUP3512X EUP3515X 3.75 EUP37506X EUP37510X EUP37512X EUP37515X 4.00 EUP4006X EUP4010X EUP4012X EUP4015X BALLOON LENGTHS 20, 25, AND 30 MM
1.50 EUP1520X — — 2.00 EUP2020X EUP2025X EUP2030X 2.25 EUP22520X EUP22525X — 2.50 EUP2520X EUP2525X EUP2530X 2.75 EUP27520X EUP27525X — 3.00 EUP3020X EUP3025X EUP3030X 3.25 EUP32520X EUP32525X — 3.50 EUP3520X EUP3525X EUP3530X 3.75 EUP37520X EUP37525X — 4.00 EUP4020X EUP4025X EUP4030X -
OCTOPUS TISSUE STABILIZER
Octopus
Tissue Stabilizers
OCTOPUS TISSUE STABILIZERS
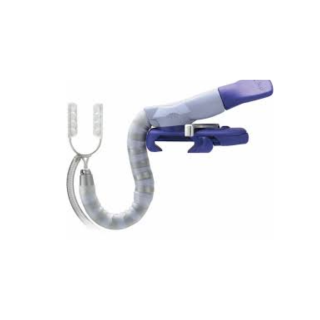
Our newest generation tissue stabilizer features automatic pod spread for effective visualization.
SUMMARY
OCTOPUS EVOLUTION AS – THE NEXT GENERATION TISSUE STABILIZER
The newest generation tissue stabiliser from Medtronic demonstrates an enduring commitment to the cardiac surgeon. With all the same features of the Octopus™ Evolution – enhanced stability, flexibility, and a lower profile for unlimited positioning options – the Octopus Evolution AS offers an automatic pod spread feature that enhances visibility at the anastomotic site.
- Automatic pod spread for effective visualisation of the anastomotic site
- Very secure arm for maximum stabilization
- Greater flexibility enables unlimited positioning options
- Simple, secure, one-handed attachment of clamp to the retractor
OCTOPUS EVOLUTION – LOW PROFILE WITH FLEXIBILITY AND REACH
The Octopus Evolution stabilizer provides enhanced stability, flexibility, and a lower profile for unlimited positioning options.
Enhanced stability and flexibility features:
- Improved visualisation with:
- Lower profile headlink
- Surgeon controlled pod-spread
- Allowance for multiple device positions including pods-up, pods-down and pods-to-the-side applications
- Increased flexibility and range of motion
- Longer, effective reach of the articulating arm provides easier access to all vessels
- Simple, secure, one-handed engagement of clamp and turret assembly
THE OCTOPUS 4.3 STABILISER FEATURES SUPERIOR ARM STRENGTH AND UNMATCHED STABILITY IN THE CLAMP/TURRET MECHANISM.
- Dual vacuum tubes offer superior tissue capture
- Internal cable enables arm twisting for maximum access and flexibility
- Reinforced arm links offer better stabilisation
- Whale tail tightens easily
- Swivel turret offers access while maximising visualisation
- Head lock design allows “toes up” position, pod spread and bend
- Rigid clamp designed to eliminate rocking
OCTOPUS 4 – EXCELLENT STABILITY AND FLEXIBILITY
The Octopus 4 tissue stabiliser includes innovative features, such as clear, malleable pods combined with a smaller diameter, more flexible arm, and an ultra-low headlink profile.
- Pod spreading optimises sewing space and stability of anastomotic site
- Reduced headlink profile improves visibility of surgical site
- Rigid, multi-material arm reduces movement at the anastomotic site
- Smaller diameter, more flexible arm offers greater flexibility in positioning
- Smaller, color-coded whale tail provides minimal obstruction
- Single vacuum tubing line minimises obstruction
- Innovative turret design increases effective arm reach, dramatically increasing positioning options with 360-degree movement
-
Everest30 INFLATION DEVICE
Everest 30
Disposable Inflation Device
OVERVIEW
Easy-to-use Rapid Inflation/Deflation Device
A simple solution for coronary balloon inflation. The Everest disposable inflation device features a quick trigger release and ergonomic pistol grip for ease of use. Plus, it’s available in your choice of 20 atm or 30 atm pressure gauge, and as a standalone or in a packaged kit.
PRODUCT DETAILS
The Everest Device Features:
- Quick trigger release so you can release pressure immediately, even at higher pressures
- Sure-grip torque knob with a ridged design for “no slip” torque control and precise pressure adjustment
- Unique ergonomic pistol grip with centered trigger that fits comfortably in either hand
- Luminescent dial face that provides accurate readability in normal and low-light conditions
EVEREST DISPOSABLE INFLATION DEVICE ORDERING INFORMATION
Disposable Inflation Device@ AC3200
Survival Kit Inflation Device@ AC305P
Everest20 inflation device
and 3-way stopcockAC2200 1 each Everest30 inflation device
and 3-way stopcockAC3200 1 each Everest20 Survival Kit: - Everest20 inflation device
- 3-way stopcock
- Piton Y-Adapter
- Metal guidewire insertion tool
- Torque handle
AC2205P 1 each Everest30 Survival Kit: - Everest30 inflation device
- 3-way stopcock
- Piton Y-Adapter
- Metal guidewire insertion tool
- Torque handle
AC3205P 1 each ACCESSORIES ORDERING INFORMATION
Metal guidewire insertion tool 006073 5 each Torque handle AC4006 5 each Piton™ Y-Adapter with metal guidewire insertion tool AC4001M 5 each Piton™ Tri-Adapter with metal guidewire insertion tool AC4002M 5 each Piton™ Y-Adapter with metal guidewire insertion tool and torque handle AC4003M 5 each -
EXPORT ADVANCE ASPIRATION CATHETER
EXPORT ADVANCE ASPIRATION CATHETER
Export Advance
Aspiration Catheter
ADVANCING PERFORMANCE
The Export Advance™ aspiration catheter delivers consistent, high-performing aspiration power when it matters most — restoring flow and protecting patients.
PRODUCT DETAILS†
Superior Deliverability1
Full-Wall Technology provides variable levels of stiffness without joints, for optimal kink resistance and catheter performance.
Dependable Delivery to the Target Site:
- Preloaded stylet enhances shaft stiffness during catheter delivery, boosting kink resistance, trackability, and pushability2
- Buddy wire compatibility allows for extra support, when needed.†
High-performing Aspiration Power:
- Large extraction lumen (0.044″ proximal; 0.043″ distal) increases aspiration power.‡
- Optimised hub geometry improves flow.‡
- Soft, short, forward-facing tip design permits excellent particle capture.†
ORDERING INFORMATION
ADVANCE 6 F min. guide I.D. 0.070″ 0.014 140 - ™Third-party brands are trademarks of their respective owners. All other brands are trademarks of a Medtronic company.
- Based on bench test data, 2013. Bench test data may not be indicative of clinical performance.
- Based on bench test data vs. Export™ AP catheter.
- Bench testing vs. Terumo Eliminate™* and Eliminate™* 3 aspiration catheters, Vascular Solutions Pronto®* LP, Pronto®* V3 and Pronto®* V4 aspiration catheters, Merit®* ASAP aspiration catheter, Kaneka Thrombuster III GR* aspiration catheter, Spectranetics QuickCat™* aspiration catheter, IHT Polska Hunter®* aspiration catheter, Medrad Possis™* Fetch™* 2 aspiration catheter, and Hexacath Recover* aspiration catheter. 10093906DOC, 2013. Bench test data may not be indicative of clinical performance.
- Based on animal studies compared with Export™ AP aspiration catheter without stylet.





Reviews
There are no reviews yet.