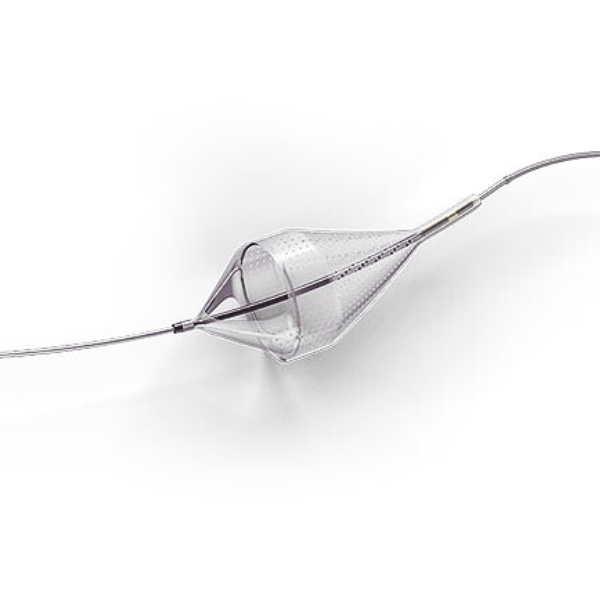
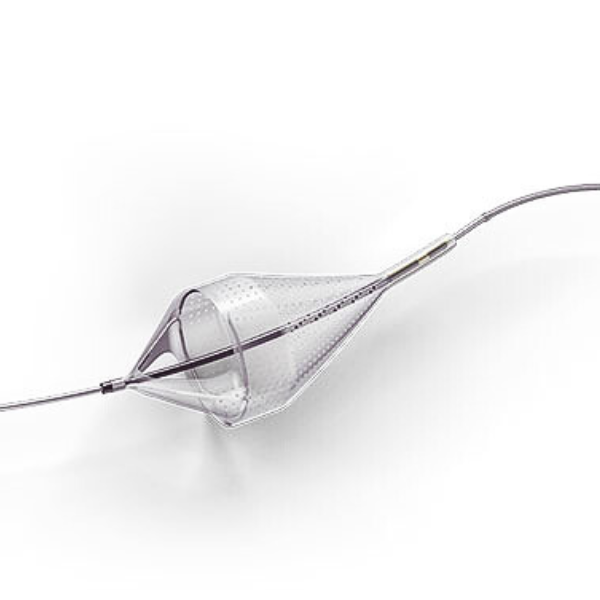
Emboshield NAV6 Embolic Protection System
Emboshield NAV6 Embolic
Indicated for Carotids and Lower Extremities
The Emboshield NAV6™ Embolic Protection System, which includes BareWire™ Filter Delivery Wires, allows the guide wire to rotate and advance freely, independent of the Emboshield NAV6™ filter.*
The Emboshield NAV6™ EPS is indicated for use as a guide wire and embolic protection system to contain and remove embolic material (thrombus / debris) while performing angioplasty and stenting procedures in carotid arteries and while performing atherectomy, during standalone procedures or together with PTA and/or stenting, in lower extremity arteries. The diameter of the artery at the site of the Filtration Element placement should be between 2.5 and 7.0 mm.
EMBOLIC PROTECTION DEVICE FOR BOTH LOWER EXTREMITY AND CAROTID ARTERIES
Captures Effectively to Minimize Downstream Complications1-3
- Centered wire design prevents bias against the vessel wall for effective debris capture.*
- Circumferential nitinol frame maintains optimal wall apposition, even on a bend.*
- Platinum-tungsten frame coils provide excellent visibility.*
- Two sizes allow for easy selection and minimal lab inventory.
Limits Filter Movement and Maintains Wire Access Through Innovative BareWire™ Design*
- The unique BareWire™ technology allows the wire to rotate and advance freely, independently of the filter.
- The filter is designed to stay in place during device delivery.
- Continued wire access, after filter is fully retracted, allows for easy delivery of additional therapy.
Navigates Skillfully Through Carotid and Lower Extremity Vasculature*
- Various types of BareWires™—distal access, workhorse, and support—are designed for various carotid anatomies.
- They promote navigational success through torturous anatomy and challenging arches.
Is Compatible with Various Atherectomy Options4
- Emboshield NAV6™ EPS and BareWire™ are compatible with a variety of atherectomy types.
- BareWire™ is available in 190 cm and 315 cm lengths.
In carotid procedures, the Emboshield NAV6™ EPS is used during stenting with the RX Acculink™ Carotid Stent System and the Xact™ Carotid Stent System.
* Data on file at Abbott.
References
- Bioangiu et al. Comparative analysis of retrieved particulate debris after superficial femoral atherectomy using three different atherectomy methods. EuroIntervention, May 2012.
- Bioangiu et al. Analysis of Retrieved Particulate Debris after Superficial Femoral Artery (SFA) Atherectomy Using the Pathway Jetstream G3 device. CCI, May 2011, 77(2) p. S57.
- Mendes et al. Clinical significance of embolic events in patients undergoing endovascular femoropopliteal interventions with or without embolic protection devices. JVS, February 2014, 59(2), 359-367.
- Philips Turbo-Elite, Medtronic TurboHawk PPES, and Boston Scientific Jetstream XC and SC. Test(s) performed by and data on file at Abbott.
IMPORTANT SAFETY INFORMATION
 Emboshield NAV6 Embolic Protection System
Emboshield NAV6 Embolic Protection System
Indications
The Emboshield NAV6 Embolic Protection System is indicated for use as a guide wire and embolic protection system to contain and remove embolic material (thrombus / debris) while performing angioplasty and stenting procedures in carotid arteries and while performing atherectomy, during standalone procedures or together with PTA and/or stenting, in lower extremity arteries. The diameter of the artery at the site of the Filtration Element placement should be between 2.5 and 7.0 mm.
Contraindications
The Emboshield NAV Embolic Protection System is contraindicated for use in
- Patients in whom anticoagulant and / or antiplatelet therapy is contraindicated.
- Patients with severe vascular tortuosity or anatomy that would preclude the safe introduction of the Guiding Catheter / Introducer Sheath, Embolic Protection System.
- Patients with a known allergy or hypersensitivity to device materials (Nitinol, Nickel, Titanium) or contrast medium, who cannot be adequately premedicated.
- Patients with uncorrected bleeding disorders.
- Lesions in the ostium of the common carotid artery.
- Inability to cross the lesion with the BareWire™ Filter Delivery Wire.
- Diffusely diseased vessels where there is no disease-free section in which to deploy the Filtration Element
- Insufficient straight section of vessel distal to the lesion to permit Filtration Element deployment.
Emboshield NAV6 Embolic Protection System
Related products
-
Glide sheath
Product overview
Expanding trans-radial access to include more patients
- Glide sheath Slender is designed with a proprietary thin-wall technology where the outside diameter is reduced by one French size while the inner-diameter equivalent is maintained.
- Glide sheath Slender is coated with Terumo’s proprietary hydrophilic coating, allowing for smooth insertion and removal.
Features and Benefits

The proprietary thin-wall technology and Terumo M Coat hydrophilic coating
- Per the American Heart Association, dedicated radial sheaths with hydrophilic coating combined with smaller diameter sheaths and a tapered tip design may reduce risk of arterial spasm and radial occlusion1
- Featuring Terumo’s proprietary hydrophilic coating, the Glide sheath Slender Sheath is the only thin-wall radial sheath on the market to demonstrate superior and consistent performance in lubricity and durability over time
The Glide sheath Slender Sheath is the only thin-wall sheath that demonstrated best in class performance in all of the American Heart Association’s identified critical features that lead to reduced spasm.
THE POWER OF TERUMO GLIDE TECHNOLOGY™: LUBRICITY OVER TIMES
Hydrophilic coating is critical to procedural success, Physicians need to trust that their sheath will be just as lubricous at the end of case as it is at the beginning.
The graph below demonstrates the results of recent benchtop testing. The lower the value, the more lubricious the coating. The straighter the line, the more consistent, and reliable, the performance.
Glide sheath Slender Sheath outperformed the other competitive sheaths in both key features: lubricity and lubricity over time.

Unmatched Clinical Evidence
- A meta-analysis of 10 clinical papers, involving 3,821 patients, demonstrated that use of Glide sheath Slender Sheath is associated with high rates of procedural success and low rates in performance related issues.
- In studies measuring patient comfort, patients who underwent percutaneous trans-radial procedures with the Glide sheath Slender Sheath reported none to low amounts of pain.
-
GUIDING CATHETER HEARTRAIL
Product Overview
IMPROVED BACK UP SUPPORT*1
Catheter designed to use contralateral wall resulting in enhanced back-up support.
UNIVERSAL SHAPE (IKARI LEFT) *2
Offer capability of accessing the right and left coronary arteries, potentially avoiding catheter exchanges.
DESIGN FOR SAFETY
Soft tip to reduce damage to the vessel wall

*1: Ikari Y, Nagaoka M, Kim JY, Moreno Y, Tanabe T. The physics of guiding catheters for the left coronary artery in transfemoral and trans-radial interventions. J Invasive Cardio. 2005 Dec; 17(12): 636-641.*2: Youssef AA, Hsieh YK, Cheng CI, We CJ. A single trans-radial guiding catheter for right and left coronary angiography and intervention. Euro intervention 2007; 3: 475-481.
Features and Benefits
Improved back up support in comparison to Terumo standard femoral guiding catheter shapes*1
 *1: Based on comparative studies made among Terumo Guiding Catheters*2: Ikari Y, Nagaoka M, Kim JY, Moreno Y, Tanabe T. The physics of guiding catheters for the left coronary artery in transfemoral and trans-radial interventions. J Invasive Cardio. 2005 Dec; 17(12): 636-641.*3: Youssef AA, Hsieh YK, Cheng CI, We CJ. A single trans-radial guiding catheter for right and left coronary angiography and intervention. Euro intervention 2007; 3: 475-481.
*1: Based on comparative studies made among Terumo Guiding Catheters*2: Ikari Y, Nagaoka M, Kim JY, Moreno Y, Tanabe T. The physics of guiding catheters for the left coronary artery in transfemoral and trans-radial interventions. J Invasive Cardio. 2005 Dec; 17(12): 636-641.*3: Youssef AA, Hsieh YK, Cheng CI, We CJ. A single trans-radial guiding catheter for right and left coronary angiography and intervention. Euro intervention 2007; 3: 475-481.General Specifications
Sizes Available Item Code JL3.5/5F TCO06GC-F5JL350N JL3.5/6F TCO06GC-F6JL350N JL4.0/6F TCO06GC-F6JL400N JR3.5/5F TCO06GC-F5JR350N JR3.5/6F TCO06GC-F6JR350N JR4.0/5F TCO06GC-F5JR400N JR4.0/6F TCO06GC-F6JR400N JR4.0/7F TCO06GC-F7JR400N MULTIPURPOSE 6F TCO06GC-F6MP010N TR4.0/6F TCO06GC-F6TR400N Heartrail II is developed to maximize your back-up force when using right and left Ikari curves during transradial interventions and through its innovative 5-in-6 system.
Its high-strength wire braiding provides exceptional kink-resistance in tortuous blood vessels, while assuring superior torque response. It also has gentle tip and powerful shaft performance.
-
RADIFOCUS INTRODUCER SHEATH
Radifocus™ Introducer II Standard Kit A
Introducer Sheath (FEMORAL)
Kit of vascular introducer with hemostatic valve with excellent blood tightness,
easy and non traumatic vessel insertion, patented valve design for a unique
features package.- Total Integrated Fit (TIF) tip tapering: tapering design at the tip of the sheath and dilator to facilitate smooth penetration
- Cross-cut hemostasis valve to avoid blood reflux and air aspiration
- Thin radiopaque sheath with anti-kinking sleeve for easy catheter handling
- Snap-on/click-off dilator lock prevents dilator back-out during insertion and allows one-hand unlocking
- Wide variety of kit variations providing all elements for quick vessel access: 4-11 Fr sheaths, 5-16 cm lengths, Surf lash needle
General specifications
Sheath length 10 cm Mini guidewire Plastic Straight and Angled
0.025″ (0.64 mm) for 4 Fr, straight mini guidewire type; 0.035″ (0.89 mm) for all others
45 cmEntry needle Plastic IV Catheter – 18G x 2 ½” (1.2 x 64 mm),
except for 4 Fr with 45 cm straight mini guidewire: 20G x 2″ (0.9 x 51 mm)
2.5 ml syringe is includedGuidewire compatibility 0.025″ (0.64 mm) for 4 Fr, straight mini guidewire type
0.035″ (0.89 mm) for all othersPackaging Tray A Kit content Sheath, dilator, plastic mini guidewire and plastic IV catheter (entry needle) Item specifications
Inner diameter Mini guidewire type 45 cm angled 45 cm straight 4 Fr RS+A40K10AQ RS+A40G10SQ 4 Fr RS*A40G10SQ RS*A40G05SQ 4 Fr RS*A40K10SQ RS*A40G07SQ 5 Fr RS*A50K10AQ RS*A50G07SQ 5 Fr RS+A50K10AQ RS+A50K10SQ 6 Fr RS+A60K10AQ RS+A60K10SQ 6 Fr RS*A60K10AQ RS*A60G07SQ 6 Fr — RS*A60G16SQZ 6 Fr — RS*A60K10SQ 6 Fr — RS+A50K10SQ 7 Fr — RS+A70K10SQ 7 Fr — RS*A70G07SQ 7 Fr — RS*A70K10SQ 8 Fr — RS+A80K10SQ 8 Fr — RS*A80K10SQ 9 Fr — RS*A90K10SQ 10 Fr — RS*A10K10SQ 11 Fr — RS*A11K10SQ A Kit includes: introducer sheath, dilator, mini Guide wire, entry venous catheter, 2.5 cc syringe
B Kit includes: introducer sheath, dilator, mini Guide wire
C Kit includes: introducer sheath, dilator
R Kit includes: introducer sheath, dilator, mini Guide wire, 18 G x 2 3/4” / 1.2 x 70 mm entry metallic needle
Other code numbers are available on special demand. For any further information, please contact your Terumo local representative
Units per box: 5 for kits in tray and 10 for kits in pouch. -
NC EUPHORA NONCOMPLIANT BALLOON CATHETER
NC Euphora™ noncompliant balloon
NC Euphora™ noncompliant balloon dilatation catheter combines Euphora technology with controlled, high-pressure performance.
Balloon Dilatation Catheter
OVERVIEW
From Collaboration to Innovation
When it comes to controlled, high-pressure performance, the NC Euphora™ noncompliant balloon dilatation catheter has what you’re looking for:
- High RBP up to 20 atm
- Low-growth profile1
- Superior deliverability1
In fact, we worked with nearly 2,000 interventional cardiologists and cath lab professionals to develop it — so you can choose it with confidence for your patients.
PRODUCT DETAILS
Looking for NC Euphora Noncompliant Performance?
ORDERING INFORMATION
BALLOON LENGTHS 6, 8, AND 12 MM
Balloon
Diameter
(mm)Balloon Length (mm) 6 8 12 2.00 NCEUP2006X NCEUP2008X NCEUP2012X 2.25 NCEUP22506X NCEUP22508X NCEUP22512X 2.50 NCEUP2506X NCEUP2508X NCEUP2512X 2.75 NCEUP27506X NCEUP27508X NCEUP27512X 3.00 NCEUP3006X NCEUP3008X NCEUP3012X 3.25 NCEUP32506X NCEUP32508X NCEUP32512X 3.50 NCEUP3506X NCEUP3508X NCEUP3512X 3.75 NCEUP37506X NCEUP37508X NCEUP37512X 4.00 NCEUP4006X NCEUP4008X NCEUP4012X 4.50 — NCEUP4508X NCEUP4512X 5.00 — NCEUP5008X NCEUP5012X BALLOON LENGTHS 15, 20, AND 27 MM
Balloon
Diameter
(mm)Balloon Length (mm) 15 20 27 2.00 NCEUP2015X NCEUP2020X — 2.25 NCEUP22515X NCEUP22520X — 2.50 NCEUP2515X NCEUP2520X NCEUP2527X 2.75 NCEUP27515X NCEUP27520X NCEUP27527X 3.00 NCEUP3015X NCEUP3020X NCEUP3027X 3.25 NCEUP32515X NCEUP32520X NCEUP32527X 3.50 NCEUP3515X NCEUP3520X NCEUP3527X 3.75 NCEUP37515X NCEUP37520X NCEUP37527X 4.00 NCEUP4015X NCEUP4020X NCEUP4027X 4.50 NCEUP4515X NCEUP4520X — 5.00 NCEUP5015X — —






Reviews
There are no reviews yet.