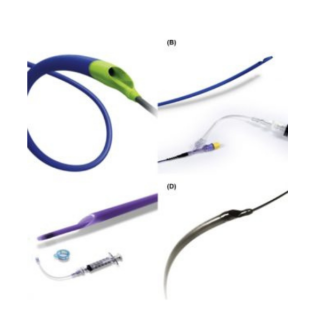
ANGIO-SEAL® (VIP Vascular Closure Device)
PRODUCT OVERVIEW

BIOABSORBABLE COMPONENTS
The device creates a mechanical seal by sandwiching the arteriotomy between a bioabsorbable anchor and collagen sponge, which dissolve in 60 to 90 days.
Three bioabsorbable components actively seal the arteriotomy:
- Anchor: Bioabsorbable co-polymer anchor placed against the inside of the vessel wall
- Collagen: Placed on top of the arteriotomy in the tissue tract
- Suture: Cinches the anchor and collagen together to form a secure seal.
LOCATE THE ARTERY
- Exchange the procedure sheath with the Angio-Seal locator system.
- Blood flow through the locator visually confirms proper sheath position in the artery.
SET THE ANCHOR
- Insert the Angio-Seal VIP device into the sheath until you hear a “click.”
- Gently pull back on the locking cap until you hear another “click.”
- The anchor is now locked in place and device is ready to be deployed.
SEAL THE PUNCTURE
- Gently pull back on the Angio-Seal VIP device until the suture has stopped spooling.
- Maintain upward tension on the device and gently advance the compaction tube until resistance is felt.
- Cut the suture and remove the device.
ANGIO-SEAL VIP PRODUCT CODES
| Part Number | French Size | Guidewire Diameter (in) |
| 610130 | 6Fr | .035 Wire |
| 610131 | 8Fr | .038 Wire |
REFERENCES
RX ONLY. Refer to the product labels and package insert for complete warnings, precautions, potential complications, and instructions for use.
- Nash JE, Evans DG. (1999). The Angio‐Seal hemostatic puncture closure device. Concept and experimental results. Herz, 24(8), 597‐606.
- Per Instructions For Use ASIN0004 revision 2018-09-01.
Indications:
The Angio-Seal Vascular Closure Device product family, including the VIP and Evolution platforms, is indicated for use in closing and reducing time to hemostasis of the femoral arterial puncture site in patients who have undergone diagnostic angiography procedures or interventional procedures using an 8 French or smaller procedural sheath for the 8 F Angio-Seal device and a 6 French or smaller procedural sheath for the 6 F Angio-Seal device. The Angio-Seal VIP and Evolution platform devices are also indicated for use to allow patients who have undergone diagnostic angiography to safely ambulate as soon as possible after sheath removal and device placement, as well as to allow patients who have undergone an interventional procedure to safely ambulate after sheath removal and device placement.
Important Safety Information:
Possible adverse events for vascular closure devices include, but are not limited to: bleeding or hematoma, AV fistula or pseudoaneurysm, infection, allergic reaction, foreign body reaction, inflammation or edema. This device should only be used by a licensed physician (or other health care professional authorized by or under the direction of such physician) possessing adequate instruction in the use of the device, e.g., participation in an Angio-Seal physician instruction program or equivalent.
ANGIO-SEAL® (VIP Vascular Closure Device)
Related products
-
PTCA BALLOON ACCUFORCE
Description
Force. Success. No Compromise.
The great balance between high pressure and high resistance, accurate dilatation and advanced deliverability. Accuforce is the latest generation of non-compliant PTCA balloons.
Characteristics
- Provides impressive high-pressure resistance up to 22 atm
- Optimizes (re)crossability and deliverability1
- Operates accurately in pre- and post-dilatation
General specifications
Shaft diameter 1.9 Fr / 0.64 mm Proximal
2.5 Fr / 0.84 mm Mid
2.6 Fr / 0.86 mm DistalUsable length 145 cm Balloon markers 2 Nominal pressure 12 atm / 1216 kPa Rated burst pressure 22 atm / 2229 kPa (2.0 mm to 4.0 mm)
20 atm / 2026 kPa (4.5 and 5.0 mm)Guidewire compatibility Maximum diameter 0.014″ / 0.36 mm Lesion entry profile 0.43 mm Coating Hydrophilic Item specifications
Ordering Information
Balloon
diameter
(mm) Balloon length (mm) / Item references 6 8 12 15 20 25 30 2.00 DC-RM2006HSW DC-RM2008HSW DC-RM2012HHW DC-RM2015HHW DC-RM2020HHW — — 2.25 DC-RM2206HSW DC-RM2208HSW DC-RM2212HHW DC-RM2215HHW DC-RM2220HHW — — 2.50 DC-RM2506HSW DC-RM2508HSW DC-RM2512HHW DC-RM2515HHW DC-RM2520HHW DC-RM2525HHW DC-RM2530HHW 2.75 DC-RM2706HSW DC-RM2708HSW DC-RM2712HHW DC-RM2715HHW DC-RM2720HHW — — 3.00 DC-RM3006HSW DC-RM3008HSW DC-RM3012HHW DC-RM3015HHW DC-RM3020HHW DC-RM3025HHW DC-RM3030HHW 3.25 DC-RM3206HSW DC-RM3208HSW DC-RM3212HHW DC-RM3215HHW DC-RM3220HHW — — 3.50 DC-RM3506HSW DC-RM3508HSW DC-RM3512HHW DC-RM3515HHW DC-RM3520HHW DC-RM3525HHW DC-RM3530HHW 3.75 DC-RM3706HSW DC-RM3708HSW DC-RM3712HHW DC-RM3715HHW DC-RM3720HHW — — 4.00 DC-RM4006HSW DC-RM4008HSW DC-RM4012HHW DC-RM4015HHW DC-RM4020HHW — — 4.50 DC-RM4506HSW DC-RM4508HSW DC-RM4512HSW DC-RM4515HSW — — — 5.00 DC-RM5006HSW DC-RM5008HSW DC-RM5012HSW DC-RM5015HSW — — — Please quote above item reference codes when placing an order.
Balloon Compliance
Balloon
diameter
(mm) Pressure (atm) 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 2.00 1.58 1.65 1.72 1.79 1.84 1.88 1.92 1.95 1.98 2.00* 2.01 2.03 2.04 2.05 2.06 2.08 2.09 2.10 2.12 2.13** 2.14 2.15 2.17 2.25 1.96 2.01 2.06 2.11 2.14 2.16 2.19 2.21 2.23 2.25* 2.26 2.28 2.29 2.31 2.32 2.34 2.35 2.36 2.38 2.39** 2.41 2.42 2.44 2.50 2.19 2.24 2.30 2.36 2.38 2.41 2.44 2.46 2.48 2.50* 2.52 2.53 2.55 2.56 2.58 2.60 2.61 2.63 2.64 2.66** 2.68 2.69 2.71 2.75 2.43 2.48 2.53 2.59 2.62 2.65 2.67 2.70 2.73 2.75* 2.77 2.79 2.80 2.82 2.84 2.86 2.87 2.89 2.91 2.93** 2.94 2.96 2.98 3.00 2.65 2.71 2.76 2.82 2.85 2.89 2.92 2.95 2.97 3.00* 3.02 3.04 3.06 3.08 3.10 3.11 3.13 3.15 3.17 3.19** 3.21 3.23 3.25 3.25 2.89 2.95 3.01 3.07 3.10 3.14 3.17 3.20 3.22 3.25* 3.27 3.29 3.31 3.33 3.35 3.37 3.39 3.42 3.44 3.46** 3.47 3.48 3.51 3.50 3.13 3.19 3.26 3.32 3.35 3.39 3.42 3.45 3.47 3.50* 3.52 3.54 3.57 3.59 3.61 3.63 3.65 3.68 3.70 3.72** 3.74 3.74 3.76 3.75 3.37 3.44 3.50 3.56 3.60 3.63 3.66 3.69 3.72 3.75* 3.77 3.80 3.82 3.84 3.86 3.88 3.90 3.92 3.94 3.96** 3.98 3.99 4.01 4.00 3.58 3.65 3.72 3.79 3.83 3.87 3.90 3.94 3.97 4.00* 4.03 4.05 4.07 4.09 4.11 4.13 4.15 4.17 4.19 4.21** 4.23 4.26 4.28 4.50 4.02 4.10 4.19 4.28 4.32 4.37 4.40 4.44 4.47 4.50* 4.53 4.56 4.59 4.63 4.66 4.68 4.71 4.75** 4.78 4.81 4.84 4.87 4.91 5.00 4.52 4.59 4.67 4.75 4.80 4.85 4.89 4.93 4.96 5.00* 5.03 5.07 5.10 5.13 5.16 5.19 5.22 5.26** 5.29 5.32 5.35 5.38 5.41 * Nominal pressure
** Rated burst pressureReferences
1 Compared with Hiryu. Non-Compliant PTCA Balloon Catheter manufactured by Terumo Corporation.
Ordering Information. Balloon diameter (mm) , Balloon length (mm) / Item references .Balloon markers: 2Usable length: 145 cmCoating: HydrophilicLesion entry profile: 0.43 mmStraight-Single Latex Plastic Terumo Accuforce Balloon Catheter, Size: Medium ; Application, Hospital ; Brand, Terumo ; Material, Latex ; Inner Diameter, 36mm. -
RESOLUTE INTEGRITY STENT
Resolute Onyx DES
for coronary artery disease
Resolute Onyx™ is a drug-eluting stent (DES) that’s different by design, optimised for complex PCI, and proven safe and effective in real-world, high bleeding risk patients on 1-month dual antiplatelet therapy (DAPT).1
Resolute Onyx DES is different by design to address your DES needs and a wide range of patient anatomies. It features:
- Best-in-class deliver ability
- Enhanced visibility with thinner struts
- Smooth side branch access
- Fast healing
- The broadest diameter range
Best-in-class deliverability
Single-wire design
Resolute Onyx DES is made from a single wire, which gives it a fluid range of motion and provides the flexibility needed for best-in-class deliverability.2

Sinusoid-formed wire

Helical wrap

Laser-fused
Competitive comparison
Resolute Onyx DES is more deliverable than laser-cut SYNERGY™* XD and XIENCE Skypoint™*, which are stiffer.
Deliverability comparison — 3.0 mm DES2
(Lower is better)

Enhanced visibility with thinner struts
Platinum-iridium core within Resolute Onyx DES increases visibility for accurate stent placement without compromising strut thickness.2Average visibility comparison2
(Higher is better)

- Cobalt alloy shell
- Platinum iridium core

Resolute Onyx cobalt alloy shell and platinum-iridum core
Smooth side branch access
Resolute Onyx DES has round struts to create a smooth passage when accessing the side branch and lower the propensity to catch during bifurcation procedures.2
Strut comparison
Rounded strut cross-section
- Resolute Onyx DES

Square strut cross-section
- SYNERGY™* DES
- XIENCE™* DES

Broadest diameter range
- Complex PCI requires a broad DES size matrix to match a wide range of patient anatomies.
- Resolute Onyx is the only DES with diameters ranging from 2.0 mm to 5.0 mm to treat a broad range of coronary vessel sizes.
Diameter (mm) Stent Length (mm) Maximum Expansion
Capabilities (MSID†) (mm)2.00 8 12 15 18 22 26 30 — — 3.50‡ 2.25 8 12 15 18 22 26 30 34 38 3.50‡ 2.50 8 12 15 18 22 26 30 34 38 3.50‡ 2.75 8 12 15 18 22 26 30 34 38 4.00‡ 3.00 8 12 15 18 22 26 30 34 38 4.00‡ 3.50 8 12 15 18 22 26 30 34 38 5.00‡ 4.00 8 12 15 18 22 26 30 34 38 5.00‡ 4.50 — 12 15 18 22 26 30 — — 6.00‡ 5.00 — 12 15 18 22 26 30 — — 6.00‡ PRODUCT DETAILS
2.00 RONYX20008X RONYX20012X RONYX20015X RONYX20018X RONYX20022X RONYX20026X RONYX20030X N/A N/A 2.25 RONYX22508X RONYX22512X RONYX22515X RONYX22518X RONYX22522X RONYX22526X RONYX22530X RONYX22534X RONYX22538X 2.50 RONYX25008X RONYX25012X RONYX25015X RONYX25018X RONYX25022X RONYX25026X RONYX25030X RONYX25034X RONYX25038X 2.75 RONYX27508X RONYX27512X RONYX27515X RONYX27518X RONYX27522X RONYX27526X RONYX27530X RONYX27534X RONYX27538X 3.00 RONYX30008X RONYX30012X RONYX30015X RONYX30018X RONYX30022X RONYX30026X RONYX30030X RONYX30034X RONYX30038X 3.50 RONYX35008X RONYX35012X RONYX35015X RONYX35015X RONYX35022X RONYX35026X RONYX35030X RONYX35034X RONYX35038X 4.00 RONYX40008X RONYX40012X RONYX40015X RONYX40018X RONYX40022X RONYX40026X RONYX40030X RONYX40034X RONYX40038X 4.50 N/A RONYX45012X RONYX45015X RONYX45018X RONYX45022X RONYX45026X RONYX45030X N/A N/A 5.00 N/A RONYX50012X RONYX50015X RONYX50018X RONYX50022X RONYX50026X RONYX5 -
EXPORT ADVANCE ASPIRATION CATHETER
EXPORT ADVANCE ASPIRATION CATHETER
Export Advance
Aspiration Catheter
ADVANCING PERFORMANCE
The Export Advance™ aspiration catheter delivers consistent, high-performing aspiration power when it matters most — restoring flow and protecting patients.
PRODUCT DETAILS†
Superior Deliverability1
Full-Wall Technology provides variable levels of stiffness without joints, for optimal kink resistance and catheter performance.
Dependable Delivery to the Target Site:
- Preloaded stylet enhances shaft stiffness during catheter delivery, boosting kink resistance, trackability, and pushability2
- Buddy wire compatibility allows for extra support, when needed.†
High-performing Aspiration Power:
- Large extraction lumen (0.044″ proximal; 0.043″ distal) increases aspiration power.‡
- Optimised hub geometry improves flow.‡
- Soft, short, forward-facing tip design permits excellent particle capture.†
ORDERING INFORMATION
ADVANCE 6 F min. guide I.D. 0.070″ 0.014 140 - ™Third-party brands are trademarks of their respective owners. All other brands are trademarks of a Medtronic company.
- Based on bench test data, 2013. Bench test data may not be indicative of clinical performance.
- Based on bench test data vs. Export™ AP catheter.
- Bench testing vs. Terumo Eliminate™* and Eliminate™* 3 aspiration catheters, Vascular Solutions Pronto®* LP, Pronto®* V3 and Pronto®* V4 aspiration catheters, Merit®* ASAP aspiration catheter, Kaneka Thrombuster III GR* aspiration catheter, Spectranetics QuickCat™* aspiration catheter, IHT Polska Hunter®* aspiration catheter, Medrad Possis™* Fetch™* 2 aspiration catheter, and Hexacath Recover* aspiration catheter. 10093906DOC, 2013. Bench test data may not be indicative of clinical performance.
- Based on animal studies compared with Export™ AP aspiration catheter without stylet.
-
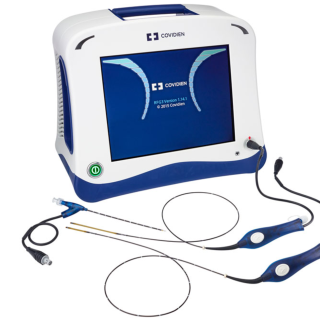
CLOSUREFAST
ClosureFast™
procedureMinimally invasive thermal
treatment for vein disease
and varicose veins.
The ClosureFast procedure uses radiofrequency energy or heat to close the diseased vein, which redirects blood flow to healthy veins, relieving symptoms.Before the procedure
You will have an ultrasound imaging exam of the leg being treated. This exam is important for assessing the diseased superficial vein and planning the procedure.
During the procedure
Your doctor will discuss the procedure with you. Here is a brief summary of what to expect:
- The ClosureFast procedure is performed on an outpatient basis. Your doctor will perform the ClosureFast procedure. Using an ultrasound, your doctor will position a catheter in the diseased vein through a small opening in the skin.
- The small catheter delivers heat to the vein wall, causing it to shrink and seal the vein. Once the diseased vein is closed, blood will reroute itself to other healthy veins.
After the procedure
- You will be taken to the recovery area to rest.
- Following the procedure, your doctor will apply a simple bandage over the insertion site and will give you compression stockings to aid in the healing process. Your doctor may encourage you to walk and to refrain from extended standing and strenuous activities for a period of time. The average patient typically resumes normal activities within a few days.1,2
- Your doctor will recommend follow-up care as needed.
-
Benefits of the ClosureFast procedure
- Relief of symptoms after two days, with a noticeable improvement in one to two week
- Minimally invasive outpatient procedure
- Less pain and bruising than laser treatment
- Faster recovery than laser treatment
- Proven results with positive patient experience
- Rapid recovery — on average, patients resume normal activities within a few days
- Lasting results — the only radiofrequency energy procedure with published long-term clinical data demonstrating safety and efficacy, with a 91.9% closure rate at five years.4
Adverse events can include nerve injury, hematoma, phlebitis, thrombosis, and/or pulmonary embolism.
Type of therapy
Thermal shrinkage of the vein wall with radiofrequency energy or heatInpatient or outpatient procedure
Outpatient procedureOne- versus two-leg treatment
Typically, each leg will be treated in separate appointmentsNeedle sticks required
One needle stick for vein access. Typically, 5 to 10 needle sticks for anesthesia.Typical post-treatment recovery
Healing of vein access site, anesthetic needle stick sites, and also healing of ablated vein sectionCompression hose
Required for approximately one weekProcedure success rate
91.9% after five years4
ClosureFast procedure FAQ
Is the ClosureFast procedure painful?
Most patients report feeling little, if any, pain during the ClosureFast procedure.3 Your doctor will give you a local or regional anesthetic to numb the treatment area.
When can I return to normal activity?
Patients treated with the ClosureFast procedure may resume normal activity more quickly than patients who undergo surgical vein stripping or laser ablation. With the ClosureFast procedure, the average patient typically resumes normal activity within a few days.2 For a few weeks following the treatment, your vein specialist may recommend a regular walking regimen and suggest you refrain from strenuous activities (heavy lifting, for example) or prolonged periods of standing.
When will my symptoms improve?
Most patients report relief of symptoms after two days, with a noticeable improvement in one to two weeks.
Is there any scarring, bruising, or swelling after the procedure?
Most patients report limited to no scarring, bruising, or swelling following the ClosureFast procedure.3
How is the ClosureFast procedure different from endovenous lasers?
Both Closure Fast and lasers use thermal technology to deliver heat into the diseased vein, but they each use a different method of delivery. Despite their similarity in using heat, a 2009 study showed that the ClosureFast procedure is associated with lower rates of pain, bruising, and complications and a faster improvement in patients’ quality of life when compared to 980 nm laser ablation.4
How is the Closure Fast procedure different from vein stripping?
During vein stripping, incisions are made in the groin and calf, and a tool is threaded through the diseased vein to pull the vein out of the leg. With the ClosureFast procedure, only one small incision is made at the insertion site and the vein is then treated and left in place. ClosureFast is a minimally invasive approach that reduces the likelihood of pain and bruising, associated with vein stripping surgery.1
Is the ClosureFast procedure covered by insurance?
Many patients have access to the ClosureFast procedure through their insurance coverage plan. Insurance companies detail access to the ClosureFast procedure and other radiofrequency ablation procedures in coverage policies for varicose veins or chronic venous insufficiency. As with all healthcare procedures, you may also have some cost associated with receiving treatment, such as a copay or coinsurance. Please reach out to your insurance company to discuss your specific plan coverage and potential costs prior to seeking treatment















Reviews
There are no reviews yet.