ANGIO-SEAL® (VIP Vascular Closure Device)
PRODUCT OVERVIEW

BIOABSORBABLE COMPONENTS
The device creates a mechanical seal by sandwiching the arteriotomy between a bioabsorbable anchor and collagen sponge, which dissolve in 60 to 90 days.
Three bioabsorbable components actively seal the arteriotomy:
- Anchor: Bioabsorbable co-polymer anchor placed against the inside of the vessel wall
- Collagen: Placed on top of the arteriotomy in the tissue tract
- Suture: Cinches the anchor and collagen together to form a secure seal.
LOCATE THE ARTERY
- Exchange the procedure sheath with the Angio-Seal locator system.
- Blood flow through the locator visually confirms proper sheath position in the artery.
SET THE ANCHOR
- Insert the Angio-Seal VIP device into the sheath until you hear a “click.”
- Gently pull back on the locking cap until you hear another “click.”
- The anchor is now locked in place and device is ready to be deployed.
SEAL THE PUNCTURE
- Gently pull back on the Angio-Seal VIP device until the suture has stopped spooling.
- Maintain upward tension on the device and gently advance the compaction tube until resistance is felt.
- Cut the suture and remove the device.
ANGIO-SEAL VIP PRODUCT CODES
| Part Number | French Size | Guidewire Diameter (in) |
| 610130 | 6Fr | .035 Wire |
| 610131 | 8Fr | .038 Wire |
REFERENCES
RX ONLY. Refer to the product labels and package insert for complete warnings, precautions, potential complications, and instructions for use.
- Nash JE, Evans DG. (1999). The Angio‐Seal hemostatic puncture closure device. Concept and experimental results. Herz, 24(8), 597‐606.
- Per Instructions For Use ASIN0004 revision 2018-09-01.
Indications:
The Angio-Seal Vascular Closure Device product family, including the VIP and Evolution platforms, is indicated for use in closing and reducing time to hemostasis of the femoral arterial puncture site in patients who have undergone diagnostic angiography procedures or interventional procedures using an 8 French or smaller procedural sheath for the 8 F Angio-Seal device and a 6 French or smaller procedural sheath for the 6 F Angio-Seal device. The Angio-Seal VIP and Evolution platform devices are also indicated for use to allow patients who have undergone diagnostic angiography to safely ambulate as soon as possible after sheath removal and device placement, as well as to allow patients who have undergone an interventional procedure to safely ambulate after sheath removal and device placement.
Important Safety Information:
Possible adverse events for vascular closure devices include, but are not limited to: bleeding or hematoma, AV fistula or pseudoaneurysm, infection, allergic reaction, foreign body reaction, inflammation or edema. This device should only be used by a licensed physician (or other health care professional authorized by or under the direction of such physician) possessing adequate instruction in the use of the device, e.g., participation in an Angio-Seal physician instruction program or equivalent.
ANGIO-SEAL® (VIP Vascular Closure Device)
Related products
-
Launcher- 8f Guiding Catheter’s
EBU@ (Extra Backup)
LA8EBU30 (EBU 3.0)
LA8EBU35 (EBU 3.5)
LA8EBU375 (EBU 3.75)
LA8EBU40 (EBU 4.0)
LA8EBU45 (EBU 4.5)
LA8EBU50 (EBU 5.0)
JL@ (Judkin Left )
LA8JL30 (JL 3.0)
LA8JL35 (JL 3.5)
LA8JL40 (JL 4.0)
LA8JL45 (JL 4.5)
LA8JL50 (JL 5.0)
LA8JL60 (JL 6.0)
JR@ (Judkin Right )
LA8JR30 (JR 3.0)
LA8JR35 (JR 3.5)
LA8JR40 (JR 4.0)
LA8JR45 (JR 4.5)
LA8JR50 (JR 5.0)
LA8JR60 (JR 6.0)
AL@ (Amplatz Left )
LA8AL75 (AL .75)
LA8AL10 (AL 1.0)
LA8AL15 (AL 1.5)
LA8AL20 (AL 2.0)
LA8AL25 (AL 2.5)
LA8AL30 (AL 3.0)
LA8AL40 (AL 4.0)
SAL@ (Short Amplatz Left)
LA8SAL75 (SAL . 75)
LA8SAL10 (SAL 1.0)
LA8SAL15 (SAL 1.5)
LA8SAL20 (SAL 2.0)
LA8SAL25 (SAL 2.5)
LA8SAL30 (SAL 3.0)
LA8SAL40 (SAL 4.0)
AR@ (Amplatz Right)
LA8AR10 (AR 1.0)
LA8AR20 (AR 2.0)
LA8ALR12 (ALR 1.2)
ECR Curves@ (Backup Support Right)
LA8ECR35 (ECR 3.5)
LA8ECR40 (ECR 4.0)
LA8ECR45 (ECR 4.5)
LA8RBU35 (RBU 3.5)
LA8RBU40 (RBU 4.0)
SCR@ (Shepherd’s Crook Right)
LA8SCR35 (SCR 3.5)
LA8SCR40 (SCR 4.0)
LA8SCR50 (SCR 5.0)
SAR@ (Short Amplatz Right)
LA8SAR10 (SAR 1.0)
LA8SAR20 (SAR 2.0)
Multipurpose@
LA8MB1 (MB 1)
LA8MB2 (MB 2)
LA8HSREL (Hockey Stick)
LA8HSI (Hockey I)
LA8HSII (Hockey II)
LA8HSIII (Hockey III)
Bypass Crafts@
LA8LCB (LCB)
LA8RCB (RCB)
LA8RCB (RCB III)
LA8RCB (RCB 90cm)
LA8IMA (IMA)
LA8IMA (IMA 90cm)
-
Everest30 INFLATION DEVICE
Everest 30
Disposable Inflation Device
OVERVIEW
Easy-to-use Rapid Inflation/Deflation Device
A simple solution for coronary balloon inflation. The Everest disposable inflation device features a quick trigger release and ergonomic pistol grip for ease of use. Plus, it’s available in your choice of 20 atm or 30 atm pressure gauge, and as a standalone or in a packaged kit.
PRODUCT DETAILS
The Everest Device Features:
- Quick trigger release so you can release pressure immediately, even at higher pressures
- Sure-grip torque knob with a ridged design for “no slip” torque control and precise pressure adjustment
- Unique ergonomic pistol grip with centered trigger that fits comfortably in either hand
- Luminescent dial face that provides accurate readability in normal and low-light conditions
EVEREST DISPOSABLE INFLATION DEVICE ORDERING INFORMATION
Disposable Inflation Device@ AC3200
Survival Kit Inflation Device@ AC305P
Everest20 inflation device
and 3-way stopcockAC2200 1 each Everest30 inflation device
and 3-way stopcockAC3200 1 each Everest20 Survival Kit: - Everest20 inflation device
- 3-way stopcock
- Piton Y-Adapter
- Metal guidewire insertion tool
- Torque handle
AC2205P 1 each Everest30 Survival Kit: - Everest30 inflation device
- 3-way stopcock
- Piton Y-Adapter
- Metal guidewire insertion tool
- Torque handle
AC3205P 1 each ACCESSORIES ORDERING INFORMATION
Metal guidewire insertion tool 006073 5 each Torque handle AC4006 5 each Piton™ Y-Adapter with metal guidewire insertion tool AC4001M 5 each Piton™ Tri-Adapter with metal guidewire insertion tool AC4002M 5 each Piton™ Y-Adapter with metal guidewire insertion tool and torque handle AC4003M 5 each -
ACT CARTRIDGE (HR-ACT)
Medtronic 402-03 ACT PLUS Disposable Test Cartridges 2-Channel Test Cartridges, High Range Activated Clotting Time (HR ACT) – Fresh whole blood test for CV surgery, PTCA, or vascular surgery, Box of 50
SKU:402-03Weight:3.00 LBSAvailability:2-3 Business daysShipping:Calculated at checkoutQuantity:Product Description
Medtronic 402-03 ACT PLUS Disposable Test Cartridges 2-Channel Test Cartridges, High Range Activated Clotting Time (HR ACT) – Fresh whole blood test for CV surgery, PTCA, or vascular surgery, Box of 50
ACT PLUS Disposable Test Cartridges 2-Channel Test Cartridges
402-01 Low Range Activated Clotting Time (LR ACT) – Fresh whole blood test for therapeutic heparin monitoring, post-angioplasty or dialysis 50 per box 402-02 Recalcified Activated Clotting Time (RACT) – For use with citrated whole blood samples at concentrations of 0.0 – 1.5 u/mL 50 per box 402-03 High Range Activated Clotting Time (HR ACT) – Fresh whole blood test for CV surgery, PTCA, or vascular surgery 50 per box 402-07 Heparinase Test Cartridge (HTC) – Fresh whole blood test used to identify the presence of heparin in the patient’s blood sample 20 per box -
FINECROSS Microcatheter
FINECROSS® MG Coronary Micro-Guide Catheter
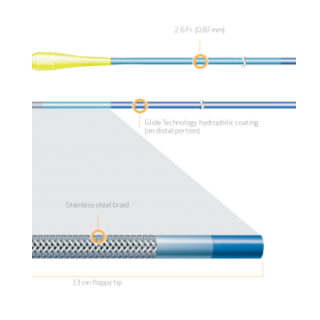
Product Code Catheter Length Distal Hydrophilic Coating Distal Outer Diameter Proximal Outer Diameter Distal Inner Diameter Proximal Inner Diameter Recommended Guidewire Size 35-1430 130cm 70cm 1.8Fr. (0.60mm) 2.6 Fr. (0.87mm) 0.018″ (0.45mm) 0.021″ (0.55mm) 0.014″ (0.36mm) 35-1450 150cm 90cm 1.8Fr. (0.60mm) 2.6 Fr. (0.87mm) 0.018″ (0.45mm) 0.021″ (0.55mm) 0.014″ (0.36mm) PRODUCT OVERVIEW
Coronary microguide catheter for integration of optimal guidewire support, superior trackability and crossability.1,2

TAPERED STAINLESS STEEL BRAID CONSTRUCTION FOR OPTIMAL GUIDEWIRE SUPPORT
FINECROSS MG stainless steel braid construction is designed to provide strength, responsiveness and support for improved pushability to access and cross complex lesions.
13cm FLOPPY DISTAL SEGMENT FOR SUPERIOR TRACKABILITY1
The distal 13 cm is ultra flexible for improved trackability around tight bends and tortuous anatomy.The floppy distal segment is designed to be atraumatic and provide an optimal balance between trackability and safety while navigating through the tortuous anatomy.1
TAPERED OUTER DIAMETER FOR SUPERIOR CROSSABILITY1,2
The outer diameter of the stainless steel shaft tapers from a proximal 2.6Fr. to a distal 1.8Fr. designed for improved crossability and guidewire handling.1,2
DOCUMENTS








Reviews
There are no reviews yet.