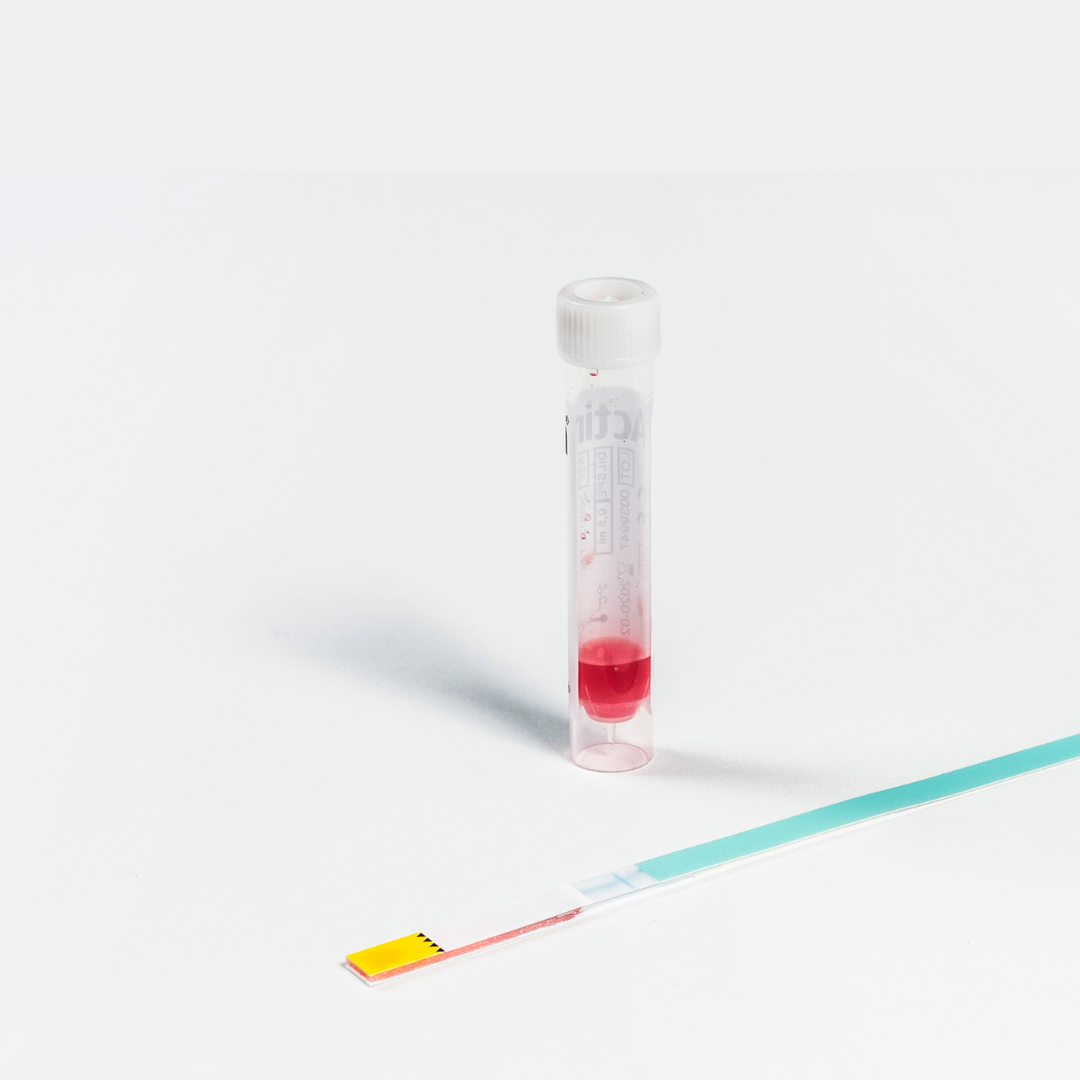
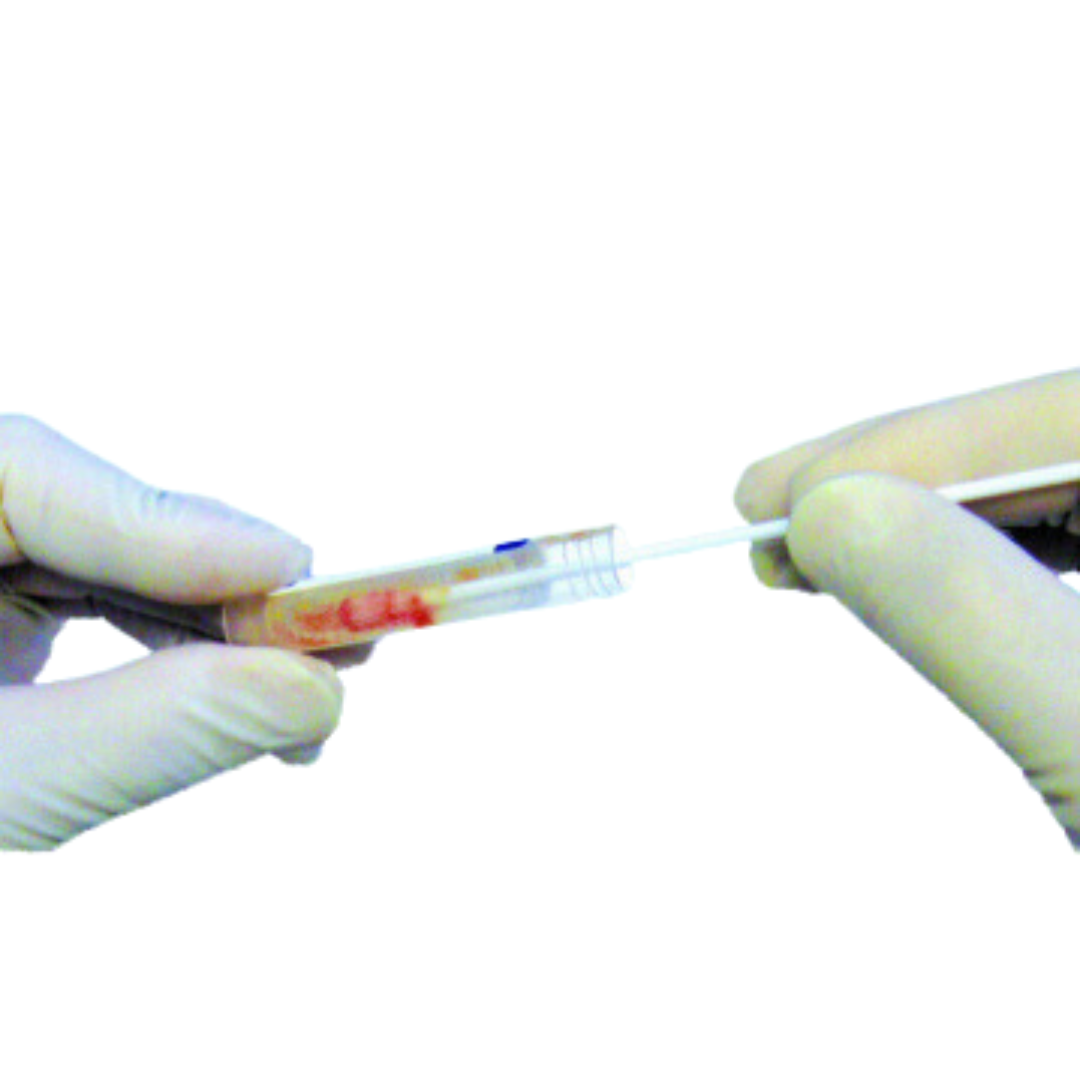
Actim PROM – Point-of-Care PROM Test
Actim PROM
A PROM test proven to be accurate even in patients with variable amounts of vaginal bleeding. Other point-of-care PROM tests are only indicated for use with trace vaginal bleeding while Actim PROM has been proven to perform with more than trace amounts of vaginal bleeding1. With up to 20% of women presenting with vaginal bleeding at the time of PROM diagnosis2, you need a reliable test for all patients. The Actim PROM advantages are clear:
- Multiple clinical studies have proven Actim PROM’s accuracy in the presence of blood
- 20 years of proven results outside the U.S. with more than 5,000,000 PROM tests performed
- Fastest POC test available, with accurate results within 5 minutes
- 1 The test has been designed to minimize interference from bleeding, but in cases of heavy bleeding the blood locally may have a higher concentration of IGFBP-1 protein. In these cases, a positive result should be interpreted with caution. Actim PROM Instructions for Use
- 2 Palacio et al.: Meta-analysis of studies on biochemical marker tests for the diagnosis of premature rupture of membranes: comparison of performance indexes. BMC Pregnancy and Childbirth 2014 14:183
Parts & Accessories
| 30831ETUS | Actim PROM Test Kit (10/Box) |
|---|---|
| 30800ETUS | Actim PROM Control Test Kit |
Related products
-
NC EUPHORA NONCOMPLIANT BALLOON CATHETER
NC Euphora™ noncompliant balloon
NC Euphora™ noncompliant balloon dilatation catheter combines Euphora technology with controlled, high-pressure performance.
Balloon Dilatation Catheter
OVERVIEW
From Collaboration to Innovation
When it comes to controlled, high-pressure performance, the NC Euphora™ noncompliant balloon dilatation catheter has what you’re looking for:
- High RBP up to 20 atm
- Low-growth profile1
- Superior deliverability1
In fact, we worked with nearly 2,000 interventional cardiologists and cath lab professionals to develop it — so you can choose it with confidence for your patients.
PRODUCT DETAILS
Looking for NC Euphora Noncompliant Performance?
ORDERING INFORMATION
BALLOON LENGTHS 6, 8, AND 12 MM
Balloon
Diameter
(mm)Balloon Length (mm) 6 8 12 2.00 NCEUP2006X NCEUP2008X NCEUP2012X 2.25 NCEUP22506X NCEUP22508X NCEUP22512X 2.50 NCEUP2506X NCEUP2508X NCEUP2512X 2.75 NCEUP27506X NCEUP27508X NCEUP27512X 3.00 NCEUP3006X NCEUP3008X NCEUP3012X 3.25 NCEUP32506X NCEUP32508X NCEUP32512X 3.50 NCEUP3506X NCEUP3508X NCEUP3512X 3.75 NCEUP37506X NCEUP37508X NCEUP37512X 4.00 NCEUP4006X NCEUP4008X NCEUP4012X 4.50 — NCEUP4508X NCEUP4512X 5.00 — NCEUP5008X NCEUP5012X BALLOON LENGTHS 15, 20, AND 27 MM
Balloon
Diameter
(mm)Balloon Length (mm) 15 20 27 2.00 NCEUP2015X NCEUP2020X — 2.25 NCEUP22515X NCEUP22520X — 2.50 NCEUP2515X NCEUP2520X NCEUP2527X 2.75 NCEUP27515X NCEUP27520X NCEUP27527X 3.00 NCEUP3015X NCEUP3020X NCEUP3027X 3.25 NCEUP32515X NCEUP32520X NCEUP32527X 3.50 NCEUP3515X NCEUP3520X NCEUP3527X 3.75 NCEUP37515X NCEUP37520X NCEUP37527X 4.00 NCEUP4015X NCEUP4020X NCEUP4027X 4.50 NCEUP4515X NCEUP4520X — 5.00 NCEUP5015X — — -
FINECROSS Microcatheter
FINECROSS® MG Coronary Micro-Guide Catheter
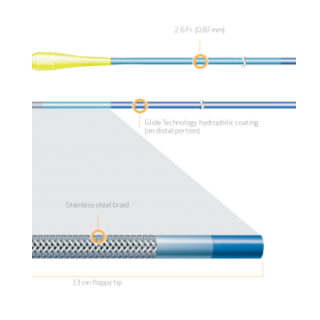
Product Code Catheter Length Distal Hydrophilic Coating Distal Outer Diameter Proximal Outer Diameter Distal Inner Diameter Proximal Inner Diameter Recommended Guidewire Size 35-1430 130cm 70cm 1.8Fr. (0.60mm) 2.6 Fr. (0.87mm) 0.018″ (0.45mm) 0.021″ (0.55mm) 0.014″ (0.36mm) 35-1450 150cm 90cm 1.8Fr. (0.60mm) 2.6 Fr. (0.87mm) 0.018″ (0.45mm) 0.021″ (0.55mm) 0.014″ (0.36mm) PRODUCT OVERVIEW
Coronary microguide catheter for integration of optimal guidewire support, superior trackability and crossability.1,2

TAPERED STAINLESS STEEL BRAID CONSTRUCTION FOR OPTIMAL GUIDEWIRE SUPPORT
FINECROSS MG stainless steel braid construction is designed to provide strength, responsiveness and support for improved pushability to access and cross complex lesions.
13cm FLOPPY DISTAL SEGMENT FOR SUPERIOR TRACKABILITY1
The distal 13 cm is ultra flexible for improved trackability around tight bends and tortuous anatomy.The floppy distal segment is designed to be atraumatic and provide an optimal balance between trackability and safety while navigating through the tortuous anatomy.1
TAPERED OUTER DIAMETER FOR SUPERIOR CROSSABILITY1,2
The outer diameter of the stainless steel shaft tapers from a proximal 2.6Fr. to a distal 1.8Fr. designed for improved crossability and guidewire handling.1,2
DOCUMENTS
-
Everest30 INFLATION DEVICE
Everest 30
Disposable Inflation Device
OVERVIEW
Easy-to-use Rapid Inflation/Deflation Device
A simple solution for coronary balloon inflation. The Everest disposable inflation device features a quick trigger release and ergonomic pistol grip for ease of use. Plus, it’s available in your choice of 20 atm or 30 atm pressure gauge, and as a standalone or in a packaged kit.
PRODUCT DETAILS
The Everest Device Features:
- Quick trigger release so you can release pressure immediately, even at higher pressures
- Sure-grip torque knob with a ridged design for “no slip” torque control and precise pressure adjustment
- Unique ergonomic pistol grip with centered trigger that fits comfortably in either hand
- Luminescent dial face that provides accurate readability in normal and low-light conditions
EVEREST DISPOSABLE INFLATION DEVICE ORDERING INFORMATION
Disposable Inflation Device@ AC3200
Survival Kit Inflation Device@ AC305P
Everest20 inflation device
and 3-way stopcockAC2200 1 each Everest30 inflation device
and 3-way stopcockAC3200 1 each Everest20 Survival Kit: - Everest20 inflation device
- 3-way stopcock
- Piton Y-Adapter
- Metal guidewire insertion tool
- Torque handle
AC2205P 1 each Everest30 Survival Kit: - Everest30 inflation device
- 3-way stopcock
- Piton Y-Adapter
- Metal guidewire insertion tool
- Torque handle
AC3205P 1 each ACCESSORIES ORDERING INFORMATION
Metal guidewire insertion tool 006073 5 each Torque handle AC4006 5 each Piton™ Y-Adapter with metal guidewire insertion tool AC4001M 5 each Piton™ Tri-Adapter with metal guidewire insertion tool AC4002M 5 each Piton™ Y-Adapter with metal guidewire insertion tool and torque handle AC4003M 5 each -
Launcher- 5f Guiding Catheter’s
EBU@ (Extra Backup)
LA5EBU30 (EBU 3.0)
LA5EBU35 (EBU 3.5)
LA5EBU375 (EBU 3.75)
LA5EBU40 (EBU 4.0)
LA5EBU45 (EBU 4.5)
LA5EBU50 (EBU 5.0)
JL@ (Judkin Left )
LA5JL30 (JL 3.0)
LA5JL35 (JL 3.5)
LA5JL40 (JL 4.0)
LA5JL45 (JL 4.5)
LA5JL50 (JL 5.0)
LA5JL60 (JL 6.0)
JR@ (Judkin Right )
LA5JR30 (JR 3.0)
LA5JR35 (JR 3.5)
LA5JR40 (JR 4.0)
LA5JR45 (JR 4.5)
LA5JR50 (JR 5.0)
LA5JR60 (JR 6.0)
AL@ (Amplatz Left )
LA5AL75 (AL .75)
LA5AL10 (AL 1.0)
LA5AL15 (AL 1.5)
LA5AL20 (AL 2.0)
LA5AL25 (AL 2.5)
LA5AL30 (AL 3.0)
LA5AL40 (AL 4.0)
SAL@ (Short Amplatz Left)
LA5SAL75 (SAL . 75)
LA5SAL10 (SAL 1.0)
LA5SAL15 (SAL 1.5)
LA5SAL20 (SAL 2.0)
LA5SAL25 (SAL 2.5)
LA5SAL30 (SAL 3.0)
LA5SAL40 (SAL 4.0)
AR@ (Amplatz Right)
LA5AR10 (AR 1.0)
LA5AR20 (AR 2.0)
LA5ALR12 (ALR 1.2)
ECR Curves@ (Backup Support Right)
LA5ECR35 (ECR 3.5)
LA5ECR40 (ECR 4.0)
LA5ECR45 (ECR 4.5)
LA5RBU35 (RBU 3.5)
LA5RBU40 (RBU 4.0)
SCR@ (Shepherd’s Crook Right)
LA5SCR35 (SCR 3.5)
LA5SCR40 (SCR 4.0)
LA5SCR50 (SCR 5.0)
SAR@ (Short Amplatz Right)
LA5SAR10 (SAR 1.0)
LA5SAR20 (SAR 2.0)
Multipurpose@
LA5MB1 (MB 1)
LA5MB2 (MB 2)
LA5HSREL (Hockey Stick)
LA5HSI (Hockey I)
LA5HSII (Hockey II)
LA5HSIII (Hockey III)
Bypass Crafts@
LA5LCB (LCB)
LA5RCB (RCB)
LA5RCB (RCB III)
LA5RCB (RCB 90cm)
LA5IMA (IMA)
LA5IMA (IMA 90cm)







Reviews
There are no reviews yet.