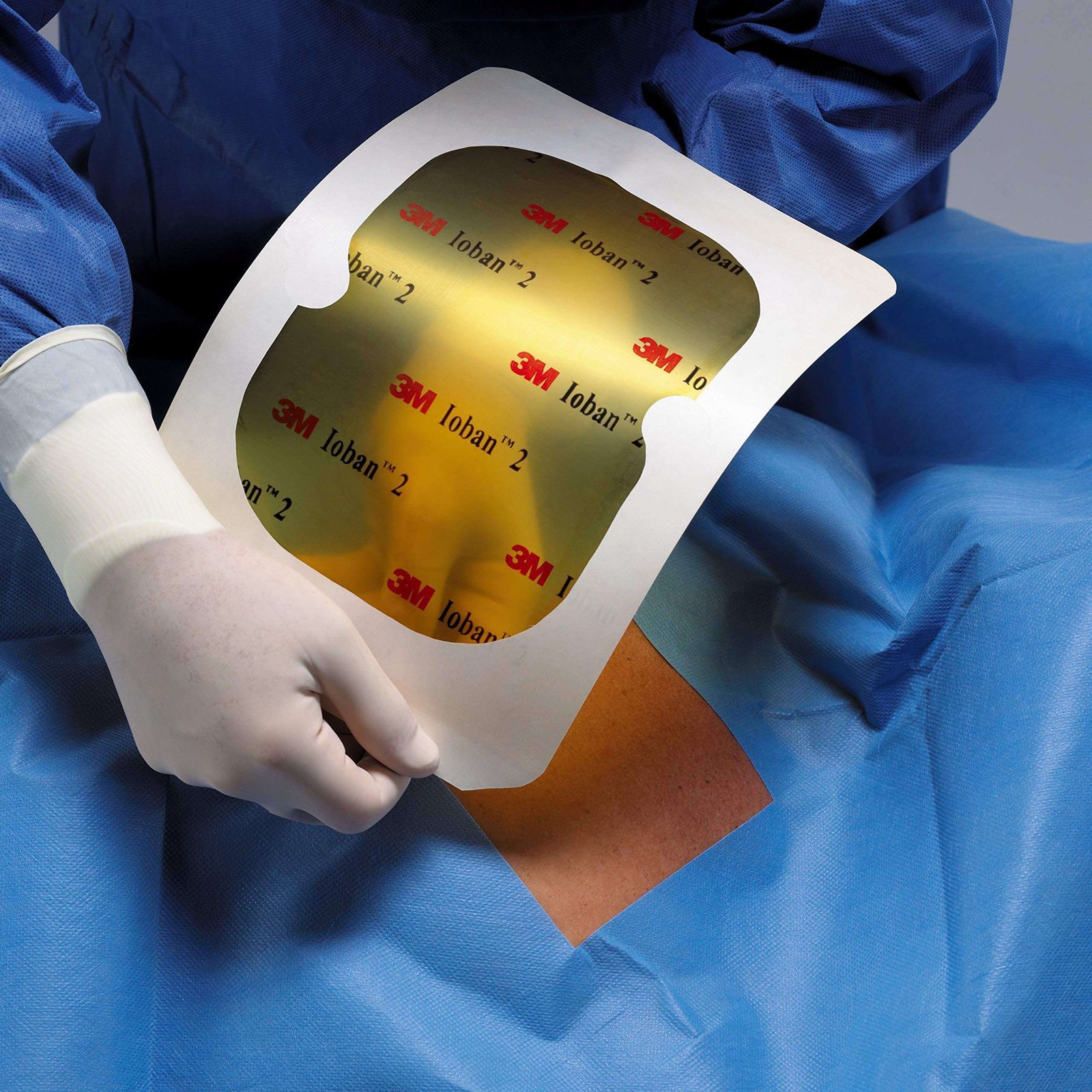
3M lOBAN 2 Antimicrobial Incise Drape ( 6650)
3M lOBAN 2
Designed with continuous broad-spectrum antimicrobial activity in the drape adhesive where it can’t be washed away
Clinically shown to help reduce the risk of wound contamination and immobilize bacteria on the skin1,2
Complies with NICE guidance on the use of incise drapes. Ioban is an iodophor impregnated drape with clinical evidence to support its performance.
Specifications
|
Brands |
Ioban™ |
|
Cladimed Code |
E51BA99 |
|
Features |
Antimicrobial |
|
Incise Area Length (metric) |
56 cm |
|
Incise Area Width (metric) |
45 cm |
|
Material |
Fabric |
|
Overall Length (Metric) |
66 cm |
|
Overall Width (Metric) |
45 cm |
|
Procedure |
General Surgery |
|
Product Type |
Incise Drapes |
|
Product Type Free Format |
Antimicrobial Incise Drape |
|
Product Use |
Incise |
|
Units per Pack |
10 |
Details
Highlights
- Designed with continuous broad-spectrum antimicrobial activity in the drape adhesive where it can’t be washed away
- Clinically shown to help reduce the risk of wound contamination and immobilize bacteria on the skin1,2
- Complies with NICE guidance on the use of incise drapes. Ioban is an iodophor impregnated drape with clinical evidence to support its performance
- High moisture-vapour transmission rate (MVTR) for breathability ensuring adhesion throughout the procedure
- Low memory stretch allows limb mobilisation or heavy retraction with reduced tension to the skin
- References 1. Dewan PA, Van Rij AM, Robinson RG, Skeggs GB, Fergus M. The use of an iodophor impregnated plastic incise drape in abdominal surgery – a controlled clinical trial. 2. French MLV, Eitzen HE, Ritter MA. The plastic surgical adhesive drape: an evaluation of its efficacy as a microbial barrier. 3. Bejko J, Tarsia V, Carrozzini M, et al. Comparison of efficacy and cost of iodine impregnated drape vs. standard drape in cardiac surgery: study in 5100 patients. 4. Rezapoor M, Tan TL, Maltenfort MG, Parvizi J. Incise draping reduces the rate of contamination of the surgical site during hip surgery: a prospective, randomized trial. 5. Alexander JW, Aerni S, Plettner JP. Development of a safe and effective one minue preoperative skin preparation
-
3M™ Ioban™ 2 Antimicrobial Incise Drapes effectively help prevent wound contamination. Evidence shows that applying an antimicrobial incise drape is a critical step in reducing the risk of wound contamination that may be associated with Surgical Site Infection (SSI).3,4 Antimicrobial Incise Drape Incise area 60cm x 45cm
Micro organisms from the patient’s skin are the most common cause of surgical site infection (SSI). Ioban 2 antimicrobial incise drapes help to reduce the risk of SSI by immobilising micro organisms and delivering antimicrobial activity at the skin surface and deeper layers.
3M lOBAN 2 Antimicrobial
Related products
-
Coronary Diagnostic Catheter (OPTITORQUE)
PRODUCT OVERVIEW
A complete line of coronary diagnostic catheters designed for superior torque control and precise placement.
RADIAL-SPECIFIC UNIVERSAL SHAPES
- Radial Specific curves designed to enable support and alignment during catheterization1
- Excellent back-up support for angiography of both RCA and LCA with a single catheter1,a
- Universal design eliminates the need for catheter exchanges and potentially shortens procedural and fluoroscopic time
1. DOF Reference
a. when compared to leading competitors
2. Ikaria article.OPTITORQUE Classic Shapes
Shape Category Product Code
5 Fr / 1.70 mmProduct Code
6 Fr / 2.00 mmShape Name (cm) Length Holes Side Tip Shape Pigtail 40-5030 40-6030 Straight Pigtail 110 6 40-5031 40-6031 Angled Pigtail 155° 110 6 Judkins Left 40-5040 40-6040 JL 3.5 (Judkins Left 3.5 cm) 100 40-5041 40-6041 JL 4.0 (Judkins Left 4.0 cm) 100 40-5042 40-6042 JL 5.0 (Judkins Left 5.0 cm) 100 Judkins Right 40-5043 40-6043 JR 3.5 (Judkins Right 3.5 cm) 100 40-5044 40-6044 JR 4.0 (Judkins Right 4.0 cm) 100 40-5045 JR 5.0 (Judkins Right 5.0 cm) 100 Bypass 40-5050 40-6050 Internal Mammary 100 Amplatz 40-5060 AL 1 (Amplatz Left Small Type) 100 40-5061 40-6061 AL 2 (Amplatz Left Middle Type) 100 UNIQUE DESIGN
- Designed for coaxial alignment to the ostium, allowing the catheter to sit at the appropriate angle for optimal engagement
- Large lumen allows high flow of contrast media and allows downsizing of French size
PROVEN PERFORMANCE
- Superior torqueability for precise placement1,a
- Excellent trackability in tortuous anatomy while navigating the radial artery1,a
- Superior flow rates for diagnostic imaging1,a
1.DOF
a. When compared to leading competitors -
PTCA Balloon Ryurei
Description
Confidence in your hands
Ryurei™ has superior crossability1 thanks to the combination of low distal profiles and Terumo’s hydrophilic M coating. The short balloon bonding and reinforced shaft1 results in enhanced trackability1 while maintaining excellent pushability1.
These features can increase physician’s confidence in success even during complex procedures.
CharacteristicsExcellent Pusability
The combination of flexible distal part, tapered core wire on the midshaft and the thick proximal hypotube, results in smooth and balanced transition across the entire catheter for high transmission force, resulting in excellent pushability
Superior Crossability2
Thanks to the combination of distal low profiles and the Terumo hydrophilic M-Coat, Ryurei offers low friction in crossing tight lesions
Enhanced Trackability2
With an optimal tip design and improved balloon bonding, Ryurei provides enhanced trackability, even in tortuous anatomy and difficult to navigate vessels.
General specifications
Shaft diameter 1.9 Fr/0.64 mm Proximal • 2.5 Fr/0.84 mm Middle • 2.4 Fr/0.79 mm to 2.7 Fr/0.89 mm (tapered) Distal (1.00 to 1.50 mm) • 2.6 Fr/0.87 mm Distal (2.00 to 4.00 mm) Usable length 145 cm Balloon markers # 1(Ø1.0 to Ø 1.5 mm) • 2(Ø2.0 to Ø4.0 mm) Nominal pressure 6 atm / 608 kPa Rated burst pressure Ø1.0 – 3.0 mm=14 atm(1419kPa) • Ø3.25 – 4.0 mm = 12 atm(1216 kPa) Guidewire compatibility Maximum diameter: 0.014″ / 0.36 mm Lesion entry profile 0.41 mm (Ø1.00 to Ø1.50 mm) • 0.43 mm (Ø2.00 to Ø4.00 mm) Coating Hydrophilic Item specifications
Balloon length / Product code
Balloon Diameter 5 mm 10 mm 15 mm 20 mm 30 mm 40 mm 1.00 mm DC-RR1005HH 1.25 mm DC-RR1205HH DC-RR1210HH DC-RR1215HH DC-RR1220HH 1.50 mm DC-RR1505HH DC-RR1510HH DC-RR1515HH DC-RR1520HH 2.00 mm DC-RR2010HHW DC-RR2015HHW DC-RR2020HHW DC-RR2030HHW DC-RR2040HHW 2.25 mm DC-RR2210HHW DC-RR2215HHW DC-RR2220HHW 2.50 mm DC-RR2510HHW DC-RR2515HHW DC-RR2520HHW DC-RR2530HHW DC-RR2540HHW 2.75 mm DCRR2710HHW DC-RR2715HHW DC-RR2720HHW 3.00 mm DC-RR3010HHW DC-RR3015HHW DC-RR3020HHW DC-RR3030HHW DC-RR3040HHW 3.25 mm DC-RR3210HHW DC-RR3215HHW DC-RR3220HHW 3.50 mm DC-RR3510HHW DC-RR3515HHW DC-RR3520HHW DC-RR3530HHW DC-RR3540HHW 3.75 mm DC-RR3710HHW DC-RR3715HHW DC-RR3720HHW 4.00 mm DC-RR4010HHW DC-RR4015HHW DC-RR4020HHW References
1 Ryurei φ1.00-5mm compared to Tazuna 1.25-10 mm- Data on file ISCD-416-31-4
* For Ryurei Ø 1.00 mm – Ø 1.50 mm.
** For Ryurei Ø 1.00mm – 5mm 2 Compared to Tazuna PTCA dilatation catheter
-
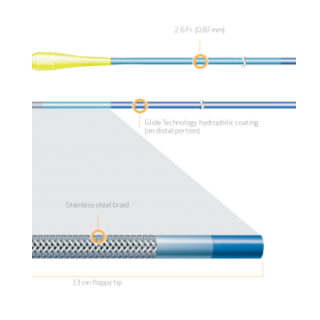
FINECROSS Microcatheter
FINECROSS® MG Coronary Micro-Guide Catheter
Product Code Catheter Length Distal Hydrophilic Coating Distal Outer Diameter Proximal Outer Diameter Distal Inner Diameter Proximal Inner Diameter Recommended Guidewire Size 35-1430 130cm 70cm 1.8Fr. (0.60mm) 2.6 Fr. (0.87mm) 0.018″ (0.45mm) 0.021″ (0.55mm) 0.014″ (0.36mm) 35-1450 150cm 90cm 1.8Fr. (0.60mm) 2.6 Fr. (0.87mm) 0.018″ (0.45mm) 0.021″ (0.55mm) 0.014″ (0.36mm) PRODUCT OVERVIEW
Coronary microguide catheter for integration of optimal guidewire support, superior trackability and crossability.1,2

TAPERED STAINLESS STEEL BRAID CONSTRUCTION FOR OPTIMAL GUIDEWIRE SUPPORT
FINECROSS MG stainless steel braid construction is designed to provide strength, responsiveness and support for improved pushability to access and cross complex lesions.
13cm FLOPPY DISTAL SEGMENT FOR SUPERIOR TRACKABILITY1
The distal 13 cm is ultra flexible for improved trackability around tight bends and tortuous anatomy.The floppy distal segment is designed to be atraumatic and provide an optimal balance between trackability and safety while navigating through the tortuous anatomy.1
TAPERED OUTER DIAMETER FOR SUPERIOR CROSSABILITY1,2
The outer diameter of the stainless steel shaft tapers from a proximal 2.6Fr. to a distal 1.8Fr. designed for improved crossability and guidewire handling.1,2
DOCUMENTS
-
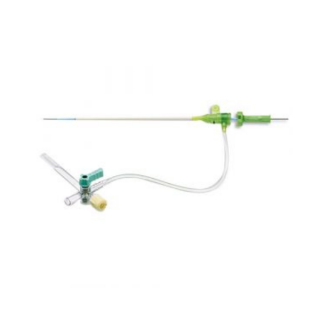
RADIFOCUS REDIAL SHEATH
Providing you the best range of Terumo Radifocus Radial Introducer Sheath and Cordis Femoral Introducer Sheath with effective & timely delivery.
The radial artery access sheath is used to gain arterial access and facilitate the insertion of catheters or other equipment for diagnostic and vascular interventions.
- The Cordis AVANTI®+ Introducer is the pioneer of catheter sheath introducer technology. Featuring a patented SLIX Valve, the AVANTI®+ introducer provides smooth transitions, monitoring capabilities and exceptional performance for your procedural success, as well as
- A hexacuspid design that provides a balance between catheter maneuverability and hemostasis.
- A rotating suture collar that facilitates procedural flexibility. It stays in place and allows patient movement.
- A kink-resistant cannula design integrating a soft, flexible inner layer with a stiffer outer layer designed to increase bendability and support, and decrease kinking.
- Atraumatic tip transitions for both the sheath and the vessel dilator are uniquely tapered and manicured. This results in smooth insertions and helps to minimize damage upon entry.
As part of the circulatory system, the radial artery supplies blood from the heart to the forearm.
Radial sheaths had the smallest OD (1 Fr smaller) when compared to similar sized conventional sheaths. For instance, the OD of 4 Fr radial sheath (1.610 ± 0.006 mm) is essentially the same as the OD of the conventional 3 Fr (1.644 ± 0.016 mm) and 3.3 Fr (1.635 ± 0.005 mm) sheaths.









Reviews
There are no reviews yet.