Emboshield NAV6 Embolic Protection System
Emboshield NAV6 Embolic
Indicated for Carotids and Lower Extremities
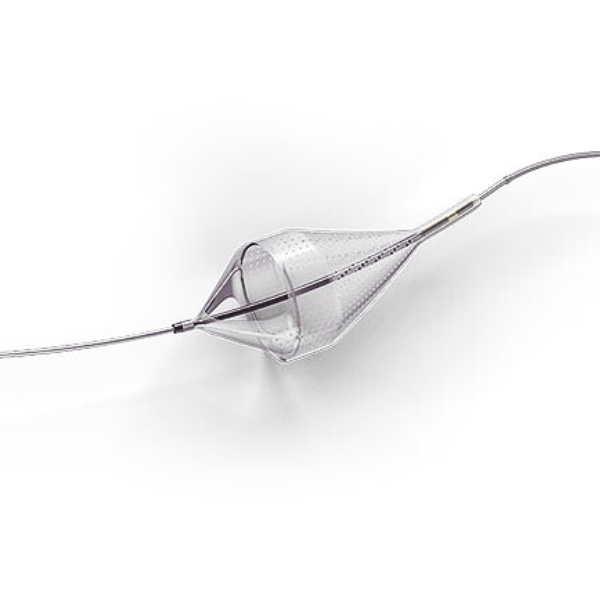
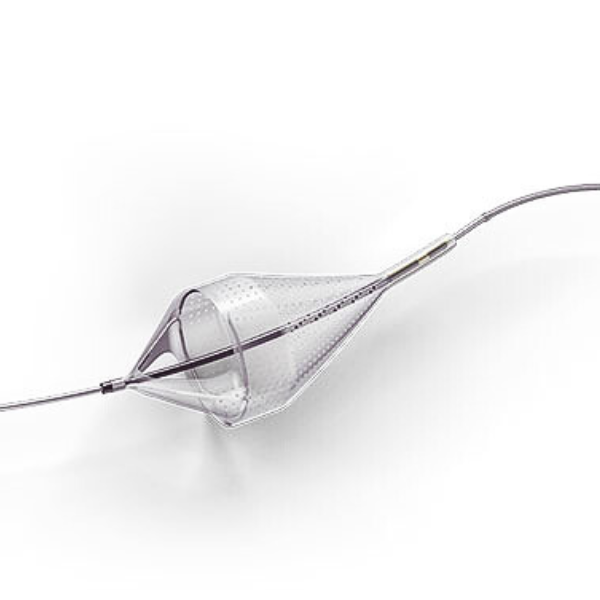
The Emboshield NAV6™ Embolic Protection System, which includes BareWire™ Filter Delivery Wires, allows the guide wire to rotate and advance freely, independent of the Emboshield NAV6™ filter.*
The Emboshield NAV6™ EPS is indicated for use as a guide wire and embolic protection system to contain and remove embolic material (thrombus / debris) while performing angioplasty and stenting procedures in carotid arteries and while performing atherectomy, during standalone procedures or together with PTA and/or stenting, in lower extremity arteries. The diameter of the artery at the site of the Filtration Element placement should be between 2.5 and 7.0 mm.
EMBOLIC PROTECTION DEVICE FOR BOTH LOWER EXTREMITY AND CAROTID ARTERIES
Captures Effectively to Minimize Downstream Complications1-3
- Centered wire design prevents bias against the vessel wall for effective debris capture.*
- Circumferential nitinol frame maintains optimal wall apposition, even on a bend.*
- Platinum-tungsten frame coils provide excellent visibility.*
- Two sizes allow for easy selection and minimal lab inventory.
Limits Filter Movement and Maintains Wire Access Through Innovative BareWire™ Design*
- The unique BareWire™ technology allows the wire to rotate and advance freely, independently of the filter.
- The filter is designed to stay in place during device delivery.
- Continued wire access, after filter is fully retracted, allows for easy delivery of additional therapy.
Navigates Skillfully Through Carotid and Lower Extremity Vasculature*
- Various types of BareWires™—distal access, workhorse, and support—are designed for various carotid anatomies.
- They promote navigational success through torturous anatomy and challenging arches.
Is Compatible with Various Atherectomy Options4
- Emboshield NAV6™ EPS and BareWire™ are compatible with a variety of atherectomy types.
- BareWire™ is available in 190 cm and 315 cm lengths.
In carotid procedures, the Emboshield NAV6™ EPS is used during stenting with the RX Acculink™ Carotid Stent System and the Xact™ Carotid Stent System.
* Data on file at Abbott.
References
- Bioangiu et al. Comparative analysis of retrieved particulate debris after superficial femoral atherectomy using three different atherectomy methods. EuroIntervention, May 2012.
- Bioangiu et al. Analysis of Retrieved Particulate Debris after Superficial Femoral Artery (SFA) Atherectomy Using the Pathway Jetstream G3 device. CCI, May 2011, 77(2) p. S57.
- Mendes et al. Clinical significance of embolic events in patients undergoing endovascular femoropopliteal interventions with or without embolic protection devices. JVS, February 2014, 59(2), 359-367.
- Philips Turbo-Elite, Medtronic TurboHawk PPES, and Boston Scientific Jetstream XC and SC. Test(s) performed by and data on file at Abbott.
IMPORTANT SAFETY INFORMATION
 Emboshield NAV6 Embolic Protection System
Emboshield NAV6 Embolic Protection System
Indications
The Emboshield NAV6 Embolic Protection System is indicated for use as a guide wire and embolic protection system to contain and remove embolic material (thrombus / debris) while performing angioplasty and stenting procedures in carotid arteries and while performing atherectomy, during standalone procedures or together with PTA and/or stenting, in lower extremity arteries. The diameter of the artery at the site of the Filtration Element placement should be between 2.5 and 7.0 mm.
Contraindications
The Emboshield NAV Embolic Protection System is contraindicated for use in
- Patients in whom anticoagulant and / or antiplatelet therapy is contraindicated.
- Patients with severe vascular tortuosity or anatomy that would preclude the safe introduction of the Guiding Catheter / Introducer Sheath, Embolic Protection System.
- Patients with a known allergy or hypersensitivity to device materials (Nitinol, Nickel, Titanium) or contrast medium, who cannot be adequately premedicated.
- Patients with uncorrected bleeding disorders.
- Lesions in the ostium of the common carotid artery.
- Inability to cross the lesion with the BareWire™ Filter Delivery Wire.
- Diffusely diseased vessels where there is no disease-free section in which to deploy the Filtration Element
- Insufficient straight section of vessel distal to the lesion to permit Filtration Element deployment.
Emboshield NAV6 Embolic Protection System
Related products
-
Launcher- 7f Guiding Catheter’s
EBU@ (Extra Backup)
LA7EBU30 (EBU 3.0)
LA7EBU35 (EBU 3.5)
LA7EBU375 (EBU 3.75)
LA7EBU40 (EBU 4.0)
LA7EBU45 (EBU 4.5)
LA7EBU50 (EBU 5.0)
JL@ (Judkin Left )
LA7JL30 (JL 3.0)
LA7JL35 (JL 3.5)
LA7JL40 (JL 4.0)
LA7JL45 (JL 4.5)
LA7JL50 (JL 5.0)
LA7JL60 (JL 6.0)
JR@ (Judkin Right )
LA7JR30 (JR 3.0)
LA7JR35 (JR 3.5)
LA7JR40 (JR 4.0)
LA7JR45 (JR 4.5)
LA7JR50 (JR 5.0)
LA7JR60 (JR 6.0)
AL@ (Amplatz Left )
LA7AL75 (AL .75)
LA7AL10 (AL 1.0)
LA7AL15 (AL 1.5)
LA7AL20 (AL 2.0)
LA7AL25 (AL 2.5)
LA7AL30 (AL 3.0)
LA7AL40 (AL 4.0)
SAL@ (Short Amplatz Left)
LA7SAL75 (SAL . 75)
LA7SAL10 (SAL 1.0)
LA7SAL15 (SAL 1.5)
LA7SAL20 (SAL 2.0)
LA7SAL25 (SAL 2.5)
LA7SAL30 (SAL 3.0)
LA7SAL40 (SAL 4.0)
AR@ (Amplatz Right)
LA7AR10 (AR 1.0)
LA7AR20 (AR 2.0)
LA7ALR12 (ALR 1.2)
ECR Curves@ (Backup Support Right)
LA7ECR35 (ECR 3.5)
LA7ECR40 (ECR 4.0)
LA7ECR45 (ECR 4.5)
LA7RBU35 (RBU 3.5)
LA7RBU40 (RBU 4.0)
SCR@ (Shepherd’s Crook Right)
LA7SCR35 (SCR 3.5)
LA7SCR40 (SCR 4.0)
LA7SCR50 (SCR 5.0)
SAR@ (Short Amplatz Right)
LA7SAR10 (SAR 1.0)
LA7SAR20 (SAR 2.0)
Multipurpose@
LA7MB1 (MB 1)
LA7MB2 (MB 2)
LA7HSREL (Hockey Stick)
LA7HSI (Hockey I)
LA7HSII (Hockey II)
LA7HSIII (Hockey III)
Bypass Crafts@
LA7LCB (LCB)
LA7RCB (RCB)
LA7RCB (RCB III)
LA7RCB (RCB 90cm)
LA7IMA (IMA)
LA7IMA (IMA 90cm)
-
Launcher- 6f Guiding Catheter’s
EBU@ (Extra Backup)
LA6EBU30 (EBU 3.0)
LA6EBU35 (EBU 3.5)
LA6EBU375 (EBU 3.75)
LA6EBU40 (EBU 4.0)
LA6EBU45 (EBU 4.5)
LA6EBU50 (EBU 5.0)
JL@ (Judkin Left )
LA6JL30 (JL 3.0)
LA6JL35 (JL 3.5)
LA6JL40 (JL 4.0)
LA6JL45 (JL 4.5)
LA6JL50 (JL 5.0)
LA6JL60 (JL 6.0)
JR@ (Judkin Right )
LA6JR30 (JR 3.0)
LA6JR35 (JR 3.5)
LA6JR40 (JR 4.0)
LA6JR45 (JR 4.5)
LA6JR50 (JR 5.0)
LA6JR60 (JR 6.0)
AL@ (Amplatz Left )
LA6AL75 (AL .75)
LA6AL10 (AL 1.0)
LA6AL15 (AL 1.5)
LA6AL20 (AL 2.0)
LA6AL25 (AL 2.5)
LA6AL30 (AL 3.0)
LA6AL40 (AL 4.0)
SAL@ (Short Amplatz Left)
LA6SAL75 (SAL . 75)
LA6SAL10 (SAL 1.0)
LA6SAL15 (SAL 1.5)
LA6SAL20 (SAL 2.0)
LA6SAL25 (SAL 2.5)
LA6SAL30 (SAL 3.0)
LA6SAL40 (SAL 4.0)
AR@ (Amplatz Right)
LA6AR10 (AR 1.0)
LA6AR20 (AR 2.0)
LA6ALR12 (ALR 1.2)
ECR Curves@ (Backup Support Right)
LA6ECR35 (ECR 3.5)
LA6ECR40 (ECR 4.0)
LA6ECR45 (ECR 4.5)
LA6RBU35 (RBU 3.5)
LA6RBU40 (RBU 4.0)
SCR@ (Shepherd’s Crook Right)
LA6SCR35 (SCR 3.5)
LA6SCR40 (SCR 4.0)
LA6SCR50 (SCR 5.0)
SAR@ (Short Amplatz Right)
LA6SAR10 (SAR 1.0)
LA6SAR20 (SAR 2.0)
Multipurpose@
LA6MB1 (MB 1)
LA6MB2 (MB 2)
LA6HSREL (Hockey Stick)
LA6HSI (Hockey I)
LA6HSII (Hockey II)
LA6HSIII (Hockey III)
Bypass Crafts@
LA6LCB (LCB)
LA6RCB (RCB)
LA6RCB (RCB III)
LA6RCB (RCB 90cm)
LA6IMA (IMA)
LA6IMA (IMA 90cm)
-
NC Sprinter PTCA BALLOON
NC Sprinter RX Noncompliant
NC Stormer over-the-wire noncompliant balloon dilatation catheter offers controlled compliance, high pressures, and comprehensive lengths.
Balloon Dilatation Catheter
OVERVIEW
The NC Sprinter™ RX noncompliant coronary balloon combines:
- Sprinter crossing technology
- High-pressure capability and controlled expansion1
ORDERING INFORMATION
BALLOON LENGTHS 6, 9, 12 MM
Balloon Diameter (mm)
Balloon Length (mm) 6 9 12 2.00 NCSP2006X NCSP2009X NCSP2012X 2.25 NCSP22506X — NCSP22512X 2.50 NCSP2506X NCSP2509X NCSP2512X 2.75 NCSP27506X NCSP27509X NCSP27512X 3.00 NCSP3006X NCSP3009X NCSP3012X 3.25 NCSP32506X NCSP32509X NCSP32512X 3.50 NCSP3506X NCSP3509X NCSP3512X 3.75 NCSP37506X NCSP37509X NCSP37512X 4.00 NCSP4006X NCSP4009X NCSP4012X 4.50 — — — 5.00 — — — BALLOON LENGTHS 15, 21, 27 MM
Balloon Diameter (mm)
Balloon Length (mm) 15 21 27 2.00 NCSP2015X NCSP2021X — 2.25 — NCSP22521X — 2.50 NCSP2515X NCSP2521X NCSP2527X 2.75 NCSP27515X NCSP27521X — 3.00 NCSP3015X NCSP3021X NCSP3027X 3.25 NCSP32515X NCSP32521X — 3.50 NCSP3515X NCSP3521X NCSP3527X 3.75 NCSP37515X NCSP37521X — 4.00 NCSP4015X NCSP4021X NCSP4027X 4.50 NCSP4515X NCSP4521X — 5.00 NCSP5015X — — -
CLOSUREFAST
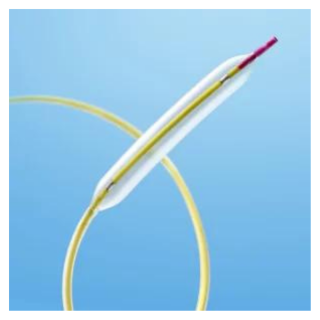
ClosureFast™
procedureMinimally invasive thermal
treatment for vein disease
and varicose veins.
The ClosureFast procedure uses radiofrequency energy or heat to close the diseased vein, which redirects blood flow to healthy veins, relieving symptoms.Before the procedure
You will have an ultrasound imaging exam of the leg being treated. This exam is important for assessing the diseased superficial vein and planning the procedure.
During the procedure
Your doctor will discuss the procedure with you. Here is a brief summary of what to expect:
- The ClosureFast procedure is performed on an outpatient basis. Your doctor will perform the ClosureFast procedure. Using an ultrasound, your doctor will position a catheter in the diseased vein through a small opening in the skin.
- The small catheter delivers heat to the vein wall, causing it to shrink and seal the vein. Once the diseased vein is closed, blood will reroute itself to other healthy veins.
After the procedure
- You will be taken to the recovery area to rest.
- Following the procedure, your doctor will apply a simple bandage over the insertion site and will give you compression stockings to aid in the healing process. Your doctor may encourage you to walk and to refrain from extended standing and strenuous activities for a period of time. The average patient typically resumes normal activities within a few days.1,2
- Your doctor will recommend follow-up care as needed.
-
Benefits of the ClosureFast procedure
- Relief of symptoms after two days, with a noticeable improvement in one to two week
- Minimally invasive outpatient procedure
- Less pain and bruising than laser treatment
- Faster recovery than laser treatment
- Proven results with positive patient experience
- Rapid recovery — on average, patients resume normal activities within a few days
- Lasting results — the only radiofrequency energy procedure with published long-term clinical data demonstrating safety and efficacy, with a 91.9% closure rate at five years.4
Adverse events can include nerve injury, hematoma, phlebitis, thrombosis, and/or pulmonary embolism.
Type of therapy
Thermal shrinkage of the vein wall with radiofrequency energy or heatInpatient or outpatient procedure
Outpatient procedureOne- versus two-leg treatment
Typically, each leg will be treated in separate appointmentsNeedle sticks required
One needle stick for vein access. Typically, 5 to 10 needle sticks for anesthesia.Typical post-treatment recovery
Healing of vein access site, anesthetic needle stick sites, and also healing of ablated vein sectionCompression hose
Required for approximately one weekProcedure success rate
91.9% after five years4
ClosureFast procedure FAQ
Is the ClosureFast procedure painful?
Most patients report feeling little, if any, pain during the ClosureFast procedure.3 Your doctor will give you a local or regional anesthetic to numb the treatment area.
When can I return to normal activity?
Patients treated with the ClosureFast procedure may resume normal activity more quickly than patients who undergo surgical vein stripping or laser ablation. With the ClosureFast procedure, the average patient typically resumes normal activity within a few days.2 For a few weeks following the treatment, your vein specialist may recommend a regular walking regimen and suggest you refrain from strenuous activities (heavy lifting, for example) or prolonged periods of standing.
When will my symptoms improve?
Most patients report relief of symptoms after two days, with a noticeable improvement in one to two weeks.
Is there any scarring, bruising, or swelling after the procedure?
Most patients report limited to no scarring, bruising, or swelling following the ClosureFast procedure.3
How is the ClosureFast procedure different from endovenous lasers?
Both Closure Fast and lasers use thermal technology to deliver heat into the diseased vein, but they each use a different method of delivery. Despite their similarity in using heat, a 2009 study showed that the ClosureFast procedure is associated with lower rates of pain, bruising, and complications and a faster improvement in patients’ quality of life when compared to 980 nm laser ablation.4
How is the Closure Fast procedure different from vein stripping?
During vein stripping, incisions are made in the groin and calf, and a tool is threaded through the diseased vein to pull the vein out of the leg. With the ClosureFast procedure, only one small incision is made at the insertion site and the vein is then treated and left in place. ClosureFast is a minimally invasive approach that reduces the likelihood of pain and bruising, associated with vein stripping surgery.1
Is the ClosureFast procedure covered by insurance?
Many patients have access to the ClosureFast procedure through their insurance coverage plan. Insurance companies detail access to the ClosureFast procedure and other radiofrequency ablation procedures in coverage policies for varicose veins or chronic venous insufficiency. As with all healthcare procedures, you may also have some cost associated with receiving treatment, such as a copay or coinsurance. Please reach out to your insurance company to discuss your specific plan coverage and potential costs prior to seeking treatment






Reviews
There are no reviews yet.